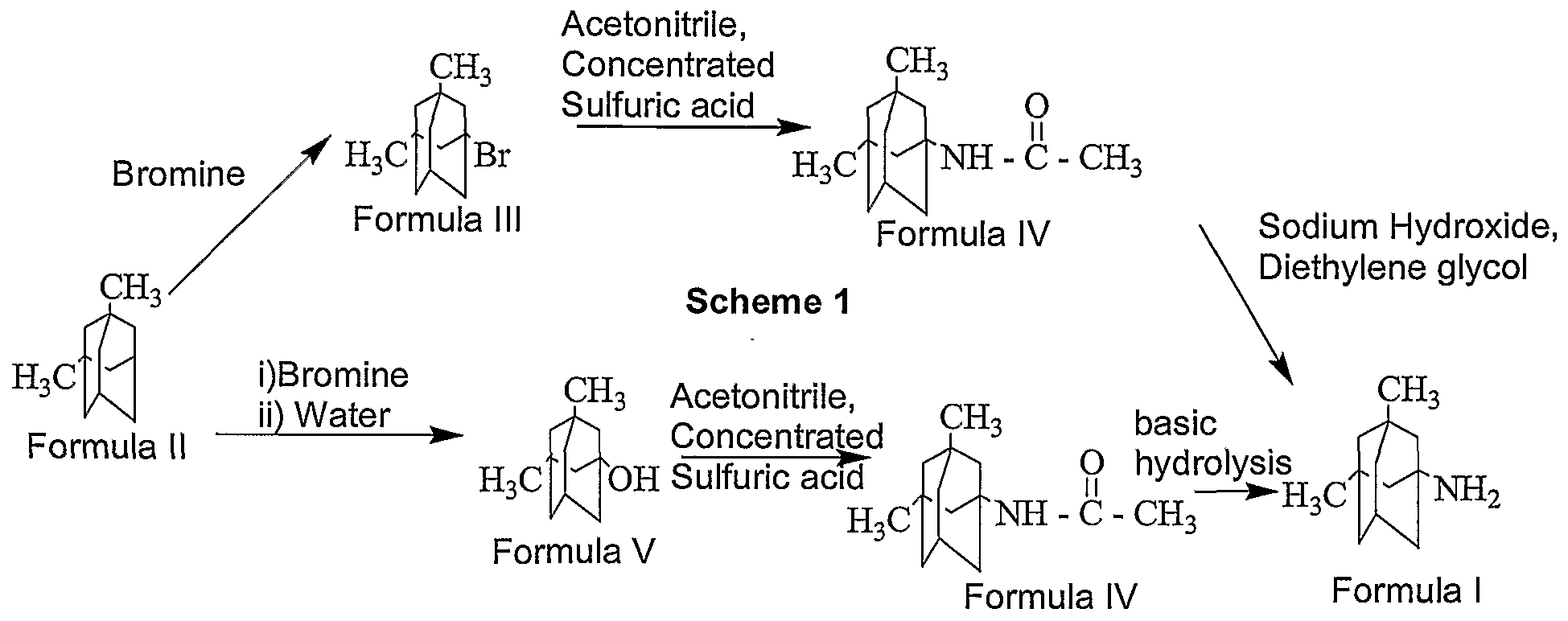
Memantine
1-amino-3,5-dimethyl adamantane
REVIEW BY ARK…..http://learnabout.arkpatentintelligence.com/drug-in-focus-memantine…… GREAT REVIEW BY
BY

Please visit www.arkpatentintelligence.com to find out more about Pipeline Patent Intelligence and how it can assist your generic drug development
Visit THEIR product page OR
Request a call to be contacted by an Ark Patent Intelligence staff member

Memantine is an orally active NMDA (N-methyl-D-aspartate) receptor antagonist which works by blocking the NMDA receptors in the brain. It blocks the excessive activity of glutamate, but still allows the normal activation of these receptors that occurs when the brain forms a memory. Therefore it improves the brain functioning in Alzheimer’s disease, and may also block the glutamate activity that could cause further damage to the brain cells.
Memantine hydrochloride is commercially available in the market in products sold under the trademark NAMENDA. It is available for oral administration as capsule shaped film-coated tablets containing 5 mg and 10 mg of memantine hydrochloride. U.S. Patent No. 3,391 ,142 discloses memantine and its related compounds, and their pharmaceutically acceptable salts.
| SynthesisReference | DrugSyn.orgUS3391142 |
|---|
……………………………….
| Country | Patent Number | Approved | Expires (estimated) |
|---|---|---|---|
| Canada | 2426492 | 2006-10-03 | 2023-05-08 |
| United States | 5061703 | 1995-04-11 | 2015-04-11 |
| Memantine is an amantadine derivative with low to moderate-affinity for NMDA receptors. It is a noncompetitive NMDA receptor antagonist that binds preferentially to NMDA receptor-operated cation channels. It blocks the effects of excessive levels of glutamate that may lead to neuronal dysfunction. It is under investigation for the treatment of Alzheimer’s disease, but there has been no clinical support for the prevention or slowing of disease progression. |

OLD CLIP
Forest Announces U.S. Availability of New Once-Daily NAMENDA XR
– Treatment for moderate to severe Alzheimer’s Disease is now available to patients in a convenient extended release formulation –
NYSE:FRX.NEW YORK–(BUSINESS WIRE)–Forest Laboratories, Inc. announced today that NAMENDA XR(TM) (memantine hydrochloride) once-daily formulation is now available in pharmacies throughout the United States. NAMENDA XR is approved by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe dementia of the Alzheimer’s type.
http://www.pharmalive.com/once-daily-namenda-xr-launched-in-us


Memantine is the first in a novel class of Alzheimer’s disease medications acting on theglutamatergic system by blocking NMDA-type glutamate receptors. It was first synthesized by Eli Lilly and Company in 1968. Memantine is marketed under the brandsAxura and Akatinol by Merz, Namenda by Forest, Ebixa and Abixa by Lundbeck andMemox by Unipharm. Memantine has been shown to have a modest effect in moderate-to-severe Alzheimer’s disease and in dementia with Lewy bodies. Despite years of research, there is little evidence of effect in mild Alzheimer’s disease. 1-4



Medical use
Memantine is approved by the U.S. F.D.A and the European Medicines Agency for treatment of moderate-to-severe Alzheimer’s disease,[5] and has now received a limited recommendation by the UK’s National Institute for Clinical Excellence for patients who fail other treatment options.[6] Within the new guidance memantine is recommended as an option for managing Alzheimer’s disease for people with: moderate Alzheimer’s disease who are intolerant of or have a contraindication to AChE (acetylcholinesterase) inhibitors or those with severe Alzheimer’s disease.
Memantine has been associated with a moderate decrease in clinical deterioration[7] with only a small positive effect on cognition, mood, behavior, and the ability to perform daily activities in moderate to severe Alzheimer’s disease.[8] There does not appear to be any benefit in mild disease.[4]
Adverse effects
Memantine is, in general, well-tolerated.[8] Common adverse drug reactions (≥1% of patients) include confusion, dizziness, drowsiness, headache, insomnia, agitation, and/or hallucinations. Less common adverse effects include vomiting, anxiety, hypertonia, cystitis, and increased libido.[7][9] It has been reported to induce reversible neurological impairment in multiple sclerosis patients, which led to the halt of an ongoing clinical trial.[10][11] Though exceedingly rare, extrapyramidal side effects (such as dystonic reactions, etc.) may occur, in particular, in the younger population.[citation needed]
A recent study demonstrates therapeutically-relevant doses of memantine in the mouse can lead to disruption of cognitive flexibility.[12]
Biochemistry
The drug belongs to a class of drugs called NMDA receptor antagonists, which reduce certain types of brain activity by binding to NMDA receptors on brain cells and blocking the activity of the neurotransmitter glutamate. At normal levels, glutamate aids in memory and learning, but if levels are too high, glutamate appears to overstimulate nerve cells, killing them through excitotoxicity.
Pharmacology
Glutamatergic (NMDA receptor)
A dysfunction of glutamatergic neurotransmission, manifested as neuronal excitotoxicity, is hypothesized to be involved in the etiology of Alzheimer’s disease. Targeting the glutamatergic system, specifically NMDA receptors, offers a novel approach to treatment in view of the limited efficacy of existing drugs targeting the cholinergic system.[13]
Memantine is a low-affinity voltage-dependent uncompetitive antagonist at glutamatergic NMDA receptors.[14][15][16][17][18] By binding to the NMDA receptor with a higher affinity than Mg2+ ions, memantine is able to inhibit the prolonged influx of Ca2+ ions, particularly from extrasynaptic receptors, which forms the basis of neuronal excitotoxicity. The low affinity, uncompetitive nature, and rapid off-rate kinetics of memantine at the level of the NMDA receptor-channel, however, preserves the function of the receptor at synapses, as it can still be activated by physiological release of glutamate following depolarization of the presynaptic neuron.[19][20][21][22][15][16][23][24][25] The interaction of memantine with NMDA receptors plays a major role in the symptomatic improvement that the drug produces in Alzheimer’s disease. Moreover, there is no evidence as yet that the ability of memantine to protect against NMDA receptor-mediated excitotoxicity has a disease-modifying effect in Alzheimer’s, although this has been suggested in animal models.[22]
Serotonergic (5-HT3 receptor)
Memantine acts as a non-competitive antagonist at the 5-HT3 receptor, with a potency similar to that for the NMDA receptor.[26] The clinical significance of this serotonergic activity in the treatment of Alzheimer’s disease is unknown.
Cholinergic (nicotinic acetylcholine receptor)
Memantine acts as a non-competitive antagonist at different neuronal nicotinic acetylcholine receptors (nAChRs) at potencies possibly similar to the NMDA and 5-HT3 receptors, but this is difficult to ascertain with accuracy because of the rapid desensitization of nAChR responses in these experiments. It can be noted that memantine is an antagonist at alpha-7 nAChR, which may contribute to initial worsening of cognitive function during early memantine treatment. Alpha-7 nAChR upregulates quickly in response to antagonism, which could explain the cognitive-enhancing effects of chronic memantine treatment.[24][27][28] It has been shown that the number of nicotinic receptors in the brain are reduced in Alzheimer’s disease, even in the absence of a general decrease in the number of neurons, and nicotinic receptor agonists are viewed as interesting targets for anti-Alzheimer drugs.[29] Consequently, this may also suggest that administration of nicotine itself may act against the effects of Alzheimer’s disease. In fact a recent small double blind placebo controlled randomized trial published in the Journal Neurology demonstrated a significant benefit from use of 15 mg nicotine patches in non-smoking elderly patients with mild cognitive impairment. The small sample sizes suggest that the effect size is substantial due to the limited statistical power inherent in a small N trial. (see : http://www.ncbi.nlm.nih.gov/pubmed/22232050)
Dopaminergic (D2 receptor)
Memantine acts as an agonist at the dopamine D2 receptor.[30]
Availability

The hydrochloride (Memantine HCl) is white water soluble powder available as capsule-shaped film-coated tablets or oral solution. The tablets are available as 5 mg, 10 mg or 20 mg of memantine hydrochloride. The oral solution contains 2 mg of memantine hydrochloride per ml.
History
Memantine was first synthesized and patented by Eli Lilly and Company in 1968 (as cited in the Merck Index), and then developed by Merz in collaboration with Neurobiological Technologies, Inc. and Children’s Hospital, Boston/Harvard Medical School, and then licensed to Forest for the U.S. and Lundbeck for selected European and international markets.
Sales of the drug reached $1.8 billion for 2010.
Research

Memantine is also being tested for generalized anxiety disorder, epilepsy, opioid dependence, systemic lupus erythematosus, depression, bipolar disorder,[31] obsessive compulsive disorder, Tourette Syndrome, problem gambling, attention-deficit hyperactivity disorder (ADHD),[32] glaucoma, tinnitus, neuropathic pain including Complex Regional Pain Syndrome,[33] pervasive developmental disorders, HIV associated dementia,[34] nystagmus,[35] multiple sclerosis,[10] autism,[36] migraine,[37] amyotrophic lateral sclerosis,[38] Down syndrome[39] and for protection of cognitive function during whole brain radiation

Memantine (1-amino-3,5-dimethyl adamantane, disclosed, e.g., in U.S. Patents No. 4,122,193; 4,273,774; 5,061 ,703) is a systemically-active uncompetitive NMDA receptor antagonist having moderate affinity for the receptor and strong voltage dependency and rapid blocking/unblocking kinetics. Memantine has been shown to be useful in alleviation of various progressive neurodegenerative disorders such as dementia in patients with moderate to severe Alzheimer’s disease, Parkinson’s disease, and spasticity (see, e.g., U. S. Patents No. 5,061 ,703; 5,614,560, and 6,034,134; Parsons et al., Neuropharmacology 1999 Jun; 38(6):735-67; Mόbius, ADAD, 1999,13:S172-178; Danysz et al., Neurotox. Res., 2000, 2:85-97; Winblad and Poritis, Int. J. Geriatr. Psychiatry, 1999, 14:135-146; Danysz et al., Cυrr. Pharm. Des., 2002, 8:835-843; Jirgensons et. al., Eur. J. Med. Chem., 2000, 35: 555-565). Memantine has also been suggested to be useful in the treatment of AIDS dementia (U.S. Patent No. 5,506,231), neuropathic pain (U.S. Patent No. 5,334,618), epilepsy, glaucoma, hepatic encephalopathy, multiple sclerosis, stroke, tardive dyskinesia (Parsons et al., 1999, supra), autism, Attention-Deficit/Hyperactivity Disorder (ADHD) and other autistic spectrum disorders (US Published Application No. 2006/0079582). Memantine is currently approved in Europe and the United States for the treatment of Alzheimer’s disease.
[0003] US Patent No. 3,391 ,142 discloses a process for the synthesis of adamantylamines, including memantine hydrochloride, involving treatment of 1-bromo- 3,5-dimethyladamantane with acetonitrile and concentrated sulfuric acid to yield the corresponding 1-acetamido-3,5-dimethyladamantane which is hydrolyzed with sodium hydroxide to yield 1-amino-3,5-dimethyladamantane which is converted to memantine hydrochloride via treatment with hydrochloric acid.
[0004] US Patent No. 4,122,193 discloses a process for the synthesis of i-amino-3,5- dialkyl adamantane derivatives, including memantine hydrochloride, which involves treatment of a 1-halo-3,5-dialkyl adamantane derivatives with a urea followed by treatment with hydrochloric acid.
R,, R2 = lower alkyl
[0005] US Patent No. 5,061 ,703 discloses a process for the synthesis of aminoadamantanes, including memantine hydrochloride, which involves halogenation and/or alkylation of the adamantane ring followed by introduction of the amino group via treatment of the halogenated derivative with formamide and subsequent hydrolysis.
HCONH2 HCI
R = H, alkyl X = Cl, Br [0006] Czech Republic Patent No. 288445 discloses a process for the synthesis of 1- amino-3,5-dimethyladannantane hydrochloride wherein 1-chloro-3,5- dimethyladamantane is reacted with formamide followed by treatment of the formamide intermediate with aqueous hydrochloric acid to yield 1-amino-3,5-dimethyladamantane hydrochloride.
HCONH2
[0007] Czech Republic Patent No. 282398 discloses a process for the synthesis of 1- amino-3,5-dimethyladamantane hydrochloride which involves treatment of 1-acetamido- 3,5-dimethyl adamantane with a base (such as postassium hydroxide) in a solvent such as methanol, ethanol, or 2-propanol.
[0008] Chinese Published Patent Publication No. CN 1566075 discloses a process for preparing 1-aminoadamantane derivatives, including memantine hydrochloride, wherein a halogenated adamantane compound (which may have substituents, including alkyl groups, in the 3 and 5 positions) is reacted with formamide or a substituted formamide, followed by deformylation under acidic conditions to yield a 1-aminoadamantane derivative.
X = Cl, Br, I R1, R2, R3 = H, alkyl, etc.
[0009] US Patent No. 5,599,998 discloses a process for the synthesis of 1- aminoadamantane derivatives, including memantine, which involves treatment of a 1- halo adamantane derivative with lithium metal to yield the lithiated intermediate which is treated with an aminating agent (such as NH2CI) under sonication conditions.
NH,CI
X = Cl, Br, I R3, R5, R7, R8 = H, F, Me
[0010] US Published Application No. 2006/025885 discloses a process for the synthesis of 1-aminoadamantane derivatives, including memantine hydrochloride, wherein a halogenated adamantane compound (which may have alkyl substituents, in the 3 and 5 positions of the adamantane ring) is reacted with acetonitrile in the presence of glacial acetic acid and concentrated sulfuric acid, followed by hydrolysis in the presence of an alkaline earth metal in a solvent such as 1-methoxy-2-propanol to yield the 1- aminoadamantane derivative which may then be converted to an acid addition salt via treatment with the appropriate acid (e.g., hydrochloric acid).
X = = F, Cl, Br, I
R1, R2 = = H, C.^alkyl
[0011] International Publication No. WO 2006/076562 discloses a process for the synthesis of memantine hydrochloride, wherein 1-halo-3,5-dimethyladamantane is reacted with acetonitrile in the presence of phosphoric acid to yield N-acetyl-1-amino- 3,5-dimethyladamantane, which may be converted to memantine (for example, via basic hydrolysis) which may then be treated with hydrochloric acid to yield memantine hydrochloride.
X = F, Cl, Br, I
[0012] International Publication No. WO 2006/122238 discloses processes for preparing memantine or an acid addition salt of memantine, which involve either reaction of 1- bromo-3,5-dimethyladamantane with formamide to form N-formyl-1-amino-3,5- dimethyladamantane or reaction of 1-hydroxy-3,5-dimethyladamantane with a hydrogen halide to obtain 1-halo-3,5-dimethyl adamantane which is then reacted with formamide to yield N-formyl-1-amino-3,5-dimethyladamantane. The N-formyl-1-amino-3,5- dimethyladamantane intermediate is deformylated under acidic conditions to yield memantine hydrochloride.
[0013] International Publication No. WO 2005/062724 discloses a process for the synthesis of 1-aminoadamantane derivatives, including memantine, which involves bromination of a compound of formula Ma, followed by hydrolysis to yield a compound of formula Md, which is treated with acetonitrile in the presence of an acid (e.g., sulfuric acid) to yield the acetamido intermediate of formula Hc. The compound of formula Mc is then hydrolyzed in the presence of acid or base to yield the aminoadamantane derivative of formula I.
R1, R2 = C1-talkyl R3 = H, C,^alkyl
[0014] International Publication No. WO 2007/101536 discloses a process for the synthesis of 1-formamido-3,5-dimethyladmantane which involves treatment of 1 ,3- dimethyladamantane with formamide in a concentrated acid, as well as a process for the conversion of 1-formamido-3,5-dimethyladmantane to memantine hydrochloride via hydrolysis with hydrochloric acid. add
[0015] Pharmaceutical active ingredients must meet strict regulatory requirements with respect to purity. Typically, products meeting such purity requirements may obtained by purifying a crude active pharmaceutical ingredient using standard purification techniques.
[0016] Memantine produced by processes known in the art may contain trace impurities including 1-amino-3,5,7-trimethyladamantane. As such trace impurities are closely related to memantine, isolating memantine from crude preparations using standard purification techniques is difficult.
[0017] Thus, a need exists to develop a process for producing memantine which is substantially free of impurities, such as 1-amino-3,5,7-trimethyladamantane.
………………………………………..
http://www.google.com/patents/EP2389351A1?cl=en
DETAILED DESCRIPTION OF THE INVENTION
[0030] A process for the preparation of 1 ,3-dimethyladamantane is shown in Scheme 1. [0031] Acenaphthene (1) is hydrogenated over a catalyst such as Raney Nickel at elevated temperature and pressure to yield perhydroacenaphthene (2). Perhydroacenaphthene is treated with a Lewis acid such as AICI3 and/or AIBr3 in the presence or absence of HCI to yield 1 ,3-dimethyladamantane which may be further purified (e.g., via fractional distillation) to provide 1 ,3-dimethyladamantane (4) containing 0.05% or less of the impurity 1,3,5-trimethyladamantane.
hydrogenation raney nickel
temp./ press.
AICL
Scheme 1 – Preparation of 1,3-dimethyladamantane
[0032] 1 ,3-dimethyladamantane containing 0.05% or less of the impurity 1 ,3,5- trimethyladamantane may be converted to memantine, or a pharmaceutically acceptable salt thereof (e.g., memantine hydrochloride), which is substantially free of the impurity 1-amino-3,5,7-trimethyladamantane, according to Scheme 2.
[0033] 1 ,3-dimethyladamantane (4), which contains 0.05% or less of the impurity 1 ,3,5- trimethyladamantane, may be treated with a halogenating agent (e.g., bromine, chlorine, or t-butylchloride) to yield 1-halo-3,5-dimethyladamantane derivative 5. Derivative 5 may be treated with formamide to yield 1-formamido-3,5- dimethyladamantane derivative 6. Alternatively, 1 ,3-dimethyladamantane, which contains 0.05% or less of the impurity 1 ,3,5-trimethyladamantane, may be treated with formamide in the presence of concentrated acid to yield 1-formamido-3,5- dimethyladamantane derivative 6. Derivative 6 may be hydrolyzed under basis or acidic conditions to provide memantine, which is substantially free of the impurity 1-amino- 3,5,7-trimethyladamantane, or a pharmaceutically acceptable salt thereof.
[0034] Derivative 5 may be also treated with acetonitrile in the presence of acid (e.g., sulfuric acid, phosphoric acid, nitric acid, a combination of acetic acid and sulfuric acid, or mixtures thereof) to yield 1-acetamido-3,5-dimethyladamantane derivative 7. Derivative 7 may be hydrolyzed to provide memantine, which is substantially free of the impurity 1-amino-3,5,7-trimethyladamantane, which may be converted to a pharmaceutically acceptable salt via treatment with a pharmaceutically acceptable acid.
Scheme 2 – Synthesis of memantine [0035] As used herein, the term halogen refers to fluorine, chlorine, bromine, and iodine.
[0036] As used herein, the term “substantially free of the impurity 1 ,3,5- trimethyladamantane” used in conjunction with 1 ,3-dimethyladamantane includes 1 ,3- dimethyladamantane which contains 0.05% or less of the impurity 1 ,3,5- trimethyladamantane.
[0037] As used herein, the term “substantially free of the impurity 1-amino-3,5,7- trimethyladamantane” used in conjunction with memantine (or a pharmaceutically acceptable salt thereof, e.g., memantine hydrochloride) includes memantine (or a pharmaceutically acceptable salt thereof, e.g., memantine hydrochloride) which contains 0.02% or less of the impurity 1-amino-3,5,7-trimethyladamantane.
Example 1
Analytical method for determining impurities in 1 ,3-dimethyladamantane
[0059] Analysis is performed on DANI 86.10 HT gas chromatograph equipped with CTC 200S automatic sampler. Millennium 3.20 software is applied for acquisition and processing of the data. All further calculations are made by MS Excel 97 software, statistics are calculated according to “Validierung in Chromotographie” Novia GmbH. 08.02.1996, Frankfurt am Main.
[0060] Gas chromatographic analysis to determine the impurity profile of 1 ,3- dimethyladamantane is performed on an SE-52 capillary column (length: 25 ; ID: 0.32 mm; df = 0.25 μm). An flame ionization detector (FID) is applied for detection. The injector temperature is 2500C and the detector temperature is also 2500C. The carrier gas is nitrogen.
[0061] Both calibration standards and analytical samples are prepared by dissolution of the components in hexane. 1-Hydroxyadamantane is applied as internal standard. All impurities are calibrated against the 1 ,3-dimethyladamantane peak.
[0062] Results for a high purity sample of 1 ,3-dimethyladamantane are shown in Table 1 below and in Figure 1.
Table 1 – Impurity profile for high purity 1 ,3-dimethyladamantane
Example 2
Preparation of 1, 3-dimethyladamantane
[0063] Acenaphthene is washed with bentonite in toluene, followed by a second wash with cyclohexene. The acenaphthene is then hydrogenated over Raney Nickel at 150 bar at a temperature of 140 to 180 0C. The catalyst is filtered off and the crude perhydroacenaphthene (240 kg) is treated with AICI3 (45 kg) and HCI (100 ml_) at 80 to 90 0C for 4 h. The reaction mixture is then heated to 120 0C for 8 h. An additional 100 ml_ of hydrochloric acid and an additional 5 kg of AICI3 are added and the reaction mixture is heated to 80 to 900C for 4 h. The crude 1 , 3-dimethyladamantane is purified via fractional distillation on a DN 300 column with oriented Sulzer type packing providing a minium of 60 theoretical plates, with the temperature profile being dependent on column pressure. The critical point is estimated on the basis of trend and is confirmed by analytical control. 1 ,3-Dimethyladamantane containing 0.05% or less of the impurity 1 ,3,5, trimethyladamantane is obtained in about 75% yield.
Example 3
Preparation of 1-bromo-3,5-dimethyladamantane
[0064] 1 , 3-dimethyladamantane containing 0.05% or less of the impurity 1 ,3,5, trimethyladamantane, as prepared in Example 2, is treated with bromine (3 equivalents) and heated to reflux for 16 h. The reaction mixture is cooled to about 15 0C and quenched with sodium bisulphite in methylene chloride. The aqueous layer is removed, and the organic layer is washed with water. The organic layer is concentrated in vacuo to yield 1-bromo-3,5-dimethyladamantane as an oil.
Example 4
Preparation of 1-amino-3,5-dimethyladamantane hydrochloride
[0065] 1-bromo-3,5-dimethyladamantane as prepared in Example 3, is treated with an excess of formamide and heated to 120 0C for 3 to 5 h. The reaction mixture is cooled and diluted with methylene chloride. This mixture is washed 4 times with a 30% sodium hydroxide solution. The organic layer is concentrated via distillation followed by addition of water. The distillation is continued to remove the organic layer, and the mixture is then cooled to below 800C. The N-formyl-1-amino-3,5-dimethyladamantane intermediate, which is optionally isolated, is then hydrolyzed by addition of a 37% hydrochloric acid solution. The reaction mixture is heated to reflux for about 3 h, and the reaction mixture is then cooled to 5 0C to yield crude 1-amino-3,5- dimethyladamantane hydrochloride which is isolated by centrifugation and washed with water followed by ethyl acetate. The crude 1-amino-3,5-dimethyladamantane hydrochloride is then reprecipitated to yield the title compound.
[0066] The purity of memantine hydrochloride prepared according to Examples 3-4 is shown in Table 2.
Table 2
Example 5
Preparation of 1-formamido-3,5-dimethyladamantane
[0067] 1,3-dimethyladamantane containing 0.05% or less of the impurity 1 ,3,5, trimethyladamantane, as prepared in Example 2, is treated with nitric acid followed by sulfuric acid at 0 0C. The reaction is stirred over night at 0 0C. The reaction mixture is poured onto 100 ml_ formamide (at 0 0C) in a round bottom flask which is equipped with a drying tube. The reaction is stirred at 0 0C for 30 min and then at room temperature for 90 min. Dichloromethane and water are then added. The organic phase is removed and washed with water and a 2 % NaHCO3-solution, dried over Na2SO4 and concentrated in vacuo. The resulting oil is purified via column chromatography to yield the title compound as a solid. Example 6
Preparation of 1-amino-3,5-dimethyladamantane
[0068] 1-formamido-3,5-dimethyladamantane, as prepared in Example 5, is hydrolyzed by addition of a 37% hydrochloric acid solution. The reaction mixture is heated to reflux for about 3 h, and the reaction mixture is then cooled to 5 0C to yield crude 1-amino-3,5- dimethyladamantane hydrochloride which is filtered and washed with water followed by ethyl acetate. The crude 1-amino-3,5-dimethyladamantane hydrochloride is then reprecipitated to yield the title compound.
Example 7
Relationship between content of the 1-amino-3,5, 7-trimethyladamantane (TMM) impurity in memantine and the 1 ,3,5-thmethyladamantane impurity in 1,3- dimethyladamantane
[0069] Memantine is synthesized according to Examples 5-6 starting from 1 ,3- Dimethyladamantane (1 ,3-DMA) spiked with different levels of the alkyl adamantane impurity 1,3,5-trimethyladamantane (TMA). The level of the corresponding aminoalkyl adamantane impurity 1-amino-3,5,7-trimethyladamantane (TMM) in the final memantine product is determined. The results (shown in Table 3) demonstrate that the amount of the 1-amino-3,5,7-trimethyladamantane (TMM) impurity in the memantine final product is dependent on the amount of 1 ,3,5-trimethyladamantane (TMA) impurity present in the 1,3-dimethyladamantane starting material.
Table 3
* * * * *


…………………………
http://www.google.com/patents/WO2006122238A1?cl=en
Memantine has the chemical name 1-amino-3,5-dimethyl adamantane (hereinafter referred to by the officially adopted name “memantine”), and can be represented by the structural Formula 1.
Formula I
Memantine is an orally active NMDA (N-methyl-D-aspartate) receptor antagonist which works by blocking the NMDA receptors in the brain. It blocks the excessive activity of glutamate, but still allows the normal activation of these receptors that occurs when the brain forms a memory. Therefore it improves the brain functioning in Alzheimer’s disease, and may also block the glutamate activity that could cause further damage to the brain cells.
Memantine hydrochloride is commercially available in the market in products sold under the trademark NAMENDA. It is available for oral administration as capsule shaped film-coated tablets containing 5 mg and 10 mg of memantine hydrochloride. U.S. Patent No. 3,391 ,142 discloses memantine and its related compounds, and their pharmaceutically acceptable salts. This patent also describes a process for the preparation of memantine as depicted in Scheme 1.
International Application Publication No. WO 2005/062724 A2 describes an alternate process for the preparation of memantine as depicted in Scheme 2, and U.S. Patent No. 5,061 ,703 discloses a process for the preparation of derivatives of memantine.
Scheme 2
A process with a reduced number of stages, and which does not require isolation of unstable or hazardous intermediates, will be helpful. Also a process which is easily scalable and is industrially feasible will be helpful.
PREPARATION OF 1-BROMO-3.5-DIMETHYL ADAMANTANE (FORMULA III)
8.0 liters of bromine was taken into a reactor and 5.1 kg of 1 ,3-dimethyl adamantane was added into the reactor slowly at 28° C in 2.5 hours. The reaction mass was maintained at 28° C for 24 hours. Reaction completion was checked using a gas chromatographic technique. After the reaction was completed, the bromine was distilled off from the reaction mass at a temperature of below 40° C. After the completion of distillation, 13.4 kg of the title compound in the form of a residue was recovered from the reactor.
Purity by GC: 98.35%,
1 ,3-dimethyl adamantane: 0.53%.
1-hydroxy-3,5-di methyl adamantane: 0.66%.
EXAMPLE 2
PREPARATION OF 1-N-FORMYL-3,5-DIMETHYL ADAMANTANE (FORMULA Vl)
64 liters of formamide was taken into a reactor and 2.55 kg of the 1-bromo- 3,5-dimethyl adamantane of Formula III obtained above was added to it. The reaction mass was heated to a temperature of 157° C and maintained for 14 hours. Reaction completion was checked using gas chromatography. After the reaction is completed, the reaction mass was cooled to 28° C. The reaction mass was then further cooled to 4° C and maintained for 4.25 hours. 37.5 liters of chilled water was added to the reaction mass slowly below 5° C. Then 37.5 liters of dichloromethane was added to the reaction mass. The temperature of the reaction mass was raised to 25° C. The reaction mass was filtered over a celite bed and the bed was washed with 12.5 liters of dichloromethane. The filtrate was taken into another reactor and stirred at 25° C for 10 minutes. The layers were separated and the aqueous layer was extracted with 15 liters of dichloromethane in 2 equal lots. The combined dichloromethane layer was washed with 51 liters of 10% sodium bicarbonate solution in 2 equal lots. The dichloromethane layer was dried over sodium sulphate and distilled under vacuum at a temperature of 40° C to yield 2.3 kg of the title compound in the form of a residue.
Purity by GC: 76.4 %,
1 ,3-dimethyl adamantane: 0.11 %, 1-hydroxy-3,5-dimethyl adamantane: 21.28%.
EXAMPLE 3
PREPARATION OF MEMANTINE HYDROCHLORIDE (FORMULA VIII): 21 liters of 36% aqueous hydrochloric acid was taken into a reactor and 2.1 kg of 1-N-formyl-3,5-dimethyl adamantane of Formula Vl obtained above was added to it. The reaction mass was heated to a temperature of 104° C and maintained for 6.5 hours. Reaction completion was checked using gas chromatographic technique. After the reaction was completed, the reaction mass was cooled to 4° C and maintained for 3 hours. The reaction mass was filtered in a centrifuge and the filtered solid was washed with 2.1 liters of chilled water. The wet cake was taken into another reactor and 8.5 liters of acetone was added to it. The reaction mass was cooled to 4° C and maintained for 3.5 hours. The reaction mass was then filtered and the filtered cake was washed with 2.1 liters of chilled acetone (chilled to a temperature of 5° C). The wet compound was dried at 73° C for 7 hours to yield 1.05 kg of the title compound. The dried compound was then milled in a micronizer (Manufacturer: Microtech Engineering company, Model: M- 50).
Purity by GC: 99.9%, 1-hydroxy-3,5-dimethyl adamantane: Less than 0.004%,
1-bromo-3,5-dimethyl adamantane: 0.01 %, 1-formamido-3,5-dimethyl adamantane: 0.03%. Residual Solvents: methanol = 231 ppm, dichloromethane = 249 ppm, cyclohexane = 43 ppm, toluene = 122 ppm. Particle size distribution: Before milling: Di0 = 3.2 μm; D50 = 18.4 μm; D90
= 96.6 μm. After milling: D10 = 1.7 μm; D50 = 9.6 μm; Dg0 = 48.3 μm.
Bulk Density: Before milling: Before tapping: 0.20 g/ml ; After tapping: 0.38 g/ml. After milling: Before tapping: 0.20 g/ml; After tapping: 0.39 g/ml. EXAMPLE 4
PREPARATION OF I-CHLORO-S.δ-DIMETHYL ADAMANTANE (FORMULA VII) 15 g of 1-hydroxy-3,5-dimethyl adamantane was taken into a round bottom flask and 300 ml of 36% aqueous hydrochloric acid was added to it. The reaction mass was stirred at 28° C for 21 hours. The reaction mass was kept for layer separation. The upper layer was separated to yield 13.0 g of the title compound in the form of crude. Purity by GC: 99.88%.
EXAMPLE 5
PREPARATION OF 1-N-FORMYL-3.5-DIMETHYL ADAMANTANE (FORMULA Vl)
10 g of 1-chloro-3,5-dimethyl adamantane of Formula VII prepared above was taken into a round bottom flask and 250 ml of formamide was added to it. The reaction mass was heated to 152° C and maintained at that temperature for 10 hours. The reaction mass was then cooled to 10 ° C, and 150 ml of water chilled to a temperature of 5° C was added to it slowly. Then 150 ml of dichloromethane was added to it and the temperature of the reaction mass was brought up to 28° C. The reaction mass was filtered through a celite bed and the celite bed was washed with 50 ml of dichloromethane. The filtrate was transferred into a separating funnel and the organic layer was separated. The aqueous layer was extracted with 150 ml of dichloromethane in two equal lots. The combined organic layer was washed with 200 ml of 10% sodium bicarbonate solution in two equal lots. The organic layer was dried over sodium sulphate and distilled under vacuum at 38° C to yield 9.6 g of the title compound. Purity by GC: 77.35%. EXAMPLE 6
PREPARATION OF 1-AMINO-3.5-DIMETHYL ADAMANTANE HYDROCHLORIDE (FORMULA VIII) 9.0 g of 1-N-formyl-3,5-dimethyl adamantane of Formula Vl and 90 ml of
36% aqueous hydrochloric acid were taken into a round bottom flask and heated to 102° C. The reaction mass was maintained at 102° C for 6 hours. The reaction mass was then allowed to cool to 2° C and maintained for 2 hours. The reaction mass was then filtered and the solid was washed with 20 ml of water chilled to a temperature of 5° C. The wet compound was taken into another round bottom flask and 54 ml of acetone was added to it. The reaction mass was stirred at 28° C for one hour, and then the reaction mass was cooled to 2° C. The reaction mass was maintained at 2° C for 2 hours. The reaction mass was then filtered and the solid was washed with 18 ml of acetone. The solid was suction dried under vacuum of 400 mm Hg and then dried in a vacuum oven at a temperature of 60° C and vacuum of 400 mm Hg for 6 hours to yield 5.0 g of the title compound. Purity by GC: 99.59%
EXAMPLE 7
PREPARATION OF 1-N-FORMYL-3.5-DIMETHYL ADAMANTANE (FORMULA VI)
13 liters of bromine was taken into a reactor and 8.3 kg of 1,3-dimethyl adamantane was added to it at 30° C. The reaction mass was maintained at 30° C for 24 hours. Reaction completion was checked using a gas chromatographic technique. 200 liters of formamide was taken in another reactor and the reaction mass from the previous reactor was added to it. The previous reactor was rinsed with 8 liters of formamide and added to the reaction mass. The temperature of the reaction mass was raised to 155° C. The reaction mass was maintained at 155° C for 8 hours. Reaction completion was checked using gas chromatography. After the reaction was completed, the reaction mass was cooled to 4° C and maintained for 1.5 hours. 125 liters of chilled water was added to the reaction mass at 4° C. 125 liters of dichloromethane was added to the reaction mass and the temperature was raised to 25° C. The reaction mass was then filtered over a celite bed and the bed was washed with 42 liters of dichloromethane. The filtrate was allowed to settle and the organic layer was separated. The aqueous layer was extracted with 124.5 liters of dichloromethane in two equal lots. The combined dichloromethane layer was washed with 166 liters of 10% aqueous solution of sodium bicarbonate in two equal lots. The dichloromethane layer was dried over sodium sulfate and distilled atmospherically to dryness at 40° C to yield 18.2 kg of the title compound.
Purity by GC: 96.29% 1 ,3-dimethyl adamantane: less than 0.004%.
1-hydroxy-3,5-dimethyl adamantane: 2.81 %.
1-bromo-3,5-dimethyl adamantane: 0.05%.
EXAMPLE 8
PREPARATION OF MEMANTINE HYDROCHLORIDE (FORMULA VIII)
24 liters of water was taken into a reactor and 24 liters of 36% aqueous hydrochloric acid was added to it. 10 kg of 1-N-formyl-3,5-dimethyl adamantane of Formula Vl was added to the reaction mass. The reaction mass was heated to a temperature of 102° C. The reaction mass was maintained at 102° C for 24 hours. Reaction completion was checked using gas chromatographic technique. After the reaction was completed the reaction mass was cooled to 5° C. The reaction mass was maintained at 5° C for 2 hours and 30 minutes and then s olids were separated using a centrifuge (Manufacturer: Nima Engineering Pvt. Ltd. Model: 14″ x 7″ – LAD CANT). The solid was washed with 10 liters of chilled water. The wet solid was then taken into another reactor and 20 liters of isopropanol was added to it. The reaction mass was stirred at 28° C for 45 minutes. Then the reaction mass was cooled to 5° C and maintained for 2.5 hours. The reaction mass was then filtered and the filtered solid was washed with 10 liters of chilled isopropanol. The wet cake was dried in an oven at 92° C for 11 hours to yield 4.6 kg of the title compound.
Purity by GC: 99.98%
1-chloro-3,5-dimethyl adamantane: less than 0.005%. 1-hydroxy-3,5-dimethyl adamantane: 0.003%.
1-N-formyl-3,5-dimethyl adamantane: 0.01%.
Residual Solvents: methanol = 33 ppm, dichloromethane = 14 ppm, acetone= 2 ppm, isopropanol = 383 ppm. Fig. 2 shows the X-ray powder diffraction pattern for the product, obtained using Cu Ka- 1 radiation (1.541 A wavelength).
EXAMPLE 9
Gas Chromatography method for memantine hydrochloride and impurities.
Chromatographic conditions:
Column: Agilent Ultra 2, or equivalent (crosslinked 5% phenylmethylsiloxane).
Length: 50 meters.
ID: 0.32 mm. Film thickness: 0.52 microns.
Column Temperature: Initial temperature: 50° C, Hold time-1: 0 minutes, Heating rate-1 : 5° C; Final temperature: 145° C, Hold time-2: 0 minutes, Heating rate-2:
10° C; Final temperature: 250° C, Final hold time: 20 minutes, Injection port temperature: 220° C, Detector temperature: 300° C. Carrier gas: Helium
Flow rate: 4.0 ml/minute
Injection mode (Split): 1 : 50
Injection volume: 1 μl.
Solvent: Hexane.
PEAK LOCATIONS:
Gas Chromatography method for residual organic solvents.
Chromatographic Conditions:
Column: Alltech AT-624 capillary, or equivalent. Length: 30 meters.
ID: 0.53 mm.
Film thickness: 3.0 μm of 6% cyanopropylphenyl-94% methylpolysiloxane.
Injector temperature: 140° C.
Detector temperature: 260° C. Mode of injection [split]: 1 :5
Carrier gas: Helium
Flow rate: 4.0 ml/minute.
Sample: 1 μl.
Diluent: N,N-dimethylacetamide. Temperature: column temperature is programmed according to the following steps: it is held at 40° C for 8 minutes then increased to 165° C at a rate of 10° C per minute and held at 165° C for 5 minutes. Again increased to 250° C at a rate of 35° C and held at 250° C for 20 minutes.

…………………………..
| WO2008062472A2 * | Oct 22, 2007 | May 29, 2008 | Cadila Healthcare Ltd | Process for the preparation of memantine |
| WO2009010806A1 * | Jul 18, 2008 | Jan 22, 2009 | Generics Uk Ltd | Assay methods for memantine |
| WO2010015415A1 * | Aug 7, 2009 | Feb 11, 2010 | Merz Pharma Gmbh & Co. Kgaa | Process for manufacturing adamantane derivatives with high yield |
| WO2010069555A1 | Dec 16, 2009 | Jun 24, 2010 | Merz Pharma Gmbh & Co. Kgaa | Method for producing memantine |
| WO2010083996A1 * | Jan 20, 2010 | Jul 29, 2010 | Merz Pharma Gmbh & Co. Kgaa | A process for preparing memantine |
| EP2555616A1 * | Apr 8, 2010 | Feb 13, 2013 | Hetero Research Foundation | Process for the preparation of memantine hydrochloride |

| WO2006122238A1 * | May 11, 2006 | Nov 16, 2006 | Mukunda Reddy Jambula | Process for preparing memantine |
| WO2007101536A1 * | Feb 20, 2007 | Sep 13, 2007 | Univ Giessen Justus Liebig | Method for producing 1-formamido-3,5-dimethyladamantane |
| WO2007126886A1 * | Mar 27, 2007 | Nov 8, 2007 | Teva Pharm Fine Chemicals Srl | Process for preparing memantine hydrochloride substantially free of impurities |
| EP1908748A1 * | Oct 5, 2006 | Apr 9, 2008 | Krka | Process for the preparation of memantine and its hydrochloric acid salt form |
References![]()
- Reisberg B, Doody R, Stöffler A, Schmitt F, Ferris S, Möbius HJ; Memantine Study Group. (2003) Memantine in moderater-to-severe Alzheimer’s disease. New Engl. J. Med. 348(14) 1333-41
- Aarsland, D; Ballard, C; Walker, Z; Bostrom, F; Alves, G; Kossakowski, K; Leroi, I; Pozo-Rodriguez, F; Minthon, L; Londos, E (July 2009). “Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial.”. Lancet neurology 8 (7): 613–8. doi:10.1016/S1474-4422(09)70146-2. PMID 19520613.
- Johansson, C; Ballard, C; Hansson, O; Palmqvist, S; Minthon, L; Aarsland, D; Londos, E (February 2011). “Efficacy of memantine in PDD and DLB: an extension study including washout and open-label treatment.”. International journal of geriatric psychiatry 26 (2): 206–13. doi:10.1002/gps.2516. PMID 20665553.
- Schneider, LS; Dagerman, KS, Higgins, JP, McShane, R (August 2011). “Lack of evidence for the efficacy of memantine in mild Alzheimer disease.”. Archives of neurology 68 (8): 991–8. doi:10.1001/archneurol.2011.69. PMID 21482915.
- Mount C, Downton C (July 2006). “Alzheimer disease: progress or profit?”. Nat Med. 12 (7): 780–4. doi:10.1038/nm0706-780. PMID 16829947.
- NICE technology appraisal January 18, 2011 Azheimer’s disease – donepezil, galantamine, rivastigmine and memantine (review): final appraisal determination
- Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006.
- Areosa SA, Sherriff F, McShane R (2005). “Memantine for dementia”. In Areosa Sastre, Almudena. Cochrane Database Syst Rev (3): CD003154. doi:10.1002/14651858.CD003154.pub4. PMID 16034889.
- Joint Formulary Committee (2004). British National Formulary (47th ed.). London: BMA and the Royal Pharmaceutical Society of Great Britain. ISBN 0-85369-584-9.
- Villoslada P, Arrondo G, Sepulcre J, Alegre M, Artieda J (December 2008). “Memantine induces reversible neurologic impairment in patients with MS”. Neurology 72 (19): 1630–3. doi:10.1212/01.wnl.0000342388.73185.80. PMID 19092106.
- Green AJ (February 2009). “Understanding pseudo. The symptoms are real, the cause is unclear”. Neurology 72 (19): 1626–7. doi:10.1212/01.wnl.0000345879.39454.68. PMID 19246422.
- Bechara J. Saab, Ruxandra M. Luca, Wing B. Yuen, Adam M. P. Saab, John C. Roder “Memantine Affects Cognitive Flexibility in the Morris Water Maze”, Journal of Alzheimer’s Disease Volume 27 Issue 3 (December 2011).
- Cacabelos R, Takeda M, Winblad B (January 1999). “The glutamatergic system and neurodegeneration in dementia: preventive strategies in Alzheimer’s disease”. Int J Geriatr Psychiatry 14 (1): 3–47. doi:10.1002/(SICI)1099-1166(199901)14:1<3::AID-GPS897>3.0.CO;2-7. PMID 10029935.
- Kornhuber J, Bormann J, Retz W, Hübers M, Riederer P (1989). “Memantine displaces [3H]MK-801 at therapeutic concentrations in postmortem human frontal cortex”. Eur.J.Pharmacol 166: 589–590. doi:10.1016/0014-2999(89)90384-1. PMID 2680528.
- Chen HSV, Pellegrini JW, Aggarwal SK, Lei SZ, Warach S, Jensen FE, Lipton SA (1 November 1992). “Open-channel block of N-methyl-D-aspartate (NMDA) responses by memantine: therapeutic advantage against NMDA receptor-mediated neurotoxicity”. J. Neurosci. 12 (11): 4427–36. PMID 1432103.
- Chen HSV, Lipton SA (15 February 1997). “Mechanism of memantine block of NMDA-activated channels in rat retinal ganglion cells: uncompetitive antagonism”. J. Physiol. (Lond.) 499 (Pt 1): 27–46. PMC 1159335. PMID 9061638.
- Rogawski, MA; Wenk GL (2003). “The neuropharmacological basis for the use of memantine in the treatment of Alzheimer’s disease”. CNS Drug Rev 9 (3): 275–308. doi:10.1111/j.1527-3458.2003.tb00254.x. PMID 14530799.
- Robinson, DM; Keating GM (2006). “Memantine: a review of its use in Alzheimer’s disease”. Drugs 66 (11): 1515–34. doi:10.2165/00003495-200666110-00015. PMID 16906789.
- Xia P, Chen HSV, Zhang D, Lipton SA (2010). “Memantine preferentially blocks extrasynaptic over synaptic NMDA receptor currents in hippocampal autapses”. J. Neurosci 30 (33): 11246–11250. doi:10.1523/JNEUROSCI.2488-10.2010. PMC 2932667. PMID 20720132.
- Kornhuber J, Weller M (1997). “Psychotogenicity and NMDA receptor antagonism: implications for neuroprotective pharmacotherapy”. Biol. Psychiatry 41 (2): 135–144. doi:10.1016/S0006-3223(96)00047-9. PMID 9018383.
- Rogawski, MA (2000). “Low affinity channel blocking (uncompetitive) NMDA receptor antagonists as therapeutic agents—toward an understanding of their favorable tolerability”. Amino Acids 19 (1): 133–49. doi:10.1007/s007260070042. PMID 11026482.
- Parsons CG, Stöffler A, Danysz W (November 2007). “Memantine: a NMDA receptor antagonist that improves memory by restoration of homeostasis in the glutamatergic system — too little activation is bad, too much is even worse”. Neuropharmacology 53 (6): 699–723. doi:10.1016/j.neuropharm.2007.07.013. PMID 17904591.
- Lipton SA (February 2006). “Paradigm shift in neuroprotection by NMDA receptor blockade: memantine and beyond”. Nature Reviews Drug Discovery 5 (2): 160–70. doi:10.1038/nrd1958. PMID 16424917.
- Chen HS, Lipton SA (June 2006). “The chemical biology of clinically tolerated NMDA receptor antagonists”. J Neurochem. 97 (6): 1611–26. doi:10.1111/j.1471-4159.2006.03991.x. PMID 16805772.
- Lipton SA (October 2007). “Pathologically activated therapeutics for neuroprotection”. Nature Reviews Neuroscience 8 (10): 803–8. doi:10.1038/nrn2229. PMID 17882256.
- Rammes G, Rupprecht R, Ferrari U, Zieglgänsberger W, Parsons CG (June 2001). “The N-methyl-D-aspartate receptor channel blockers memantine, MRZ 2/579 and other amino-alkyl-cyclohexanes antagonise 5-HT(3) receptor currents in cultured HEK-293 and N1E-115 cell systems in a non-competitive manner”. Neurosci Lett. 306 (1–2): 81–4. doi:10.1016/S0304-3940(01)01872-9. PMID 11403963.
- Buisson B, Bertrand D (1 March 1998). “Open-channel blockers at the human alpha4beta2 neuronal nicotinic acetylcholine receptor”. Mol Pharmacol. 53 (3): 555–63. PMID 9495824.
- Aracava Y, Pereira EF, Maelicke A, Albuquerque EX (March 2005). “Memantine blocks alpha7* nicotinic acetylcholine receptors more potently than n-methyl-D-aspartate receptors in rat hippocampal neurons”. J Pharmacol Exp Ther. 312 (3): 1195–205. doi:10.1124/jpet.104.077172. PMID 15522999.
- Gotti C, Clementi F (December 2004). “Neuronal nicotinic receptors: from structure to pathology”. Prog Neurobiol. 74 (6): 363–96. doi:10.1016/j.pneurobio.2004.09.006. PMID 15649582.
- Seeman P, Caruso C, Lasaga M (February 2008). “Memantine agonist action at dopamine D2High receptors”. Synapse 62 (2): 149–53. doi:10.1002/syn.20472. PMID 18000814.
- http://www.psycheducation.org/depression/meds/memantine.htm
- Open-Label Pilot Study of Namenda in Adult Subjects With ADHD and ADHD NOS [1]
- Dan Ziegler. “New drugs to prevent or treat diabetic polyneuropathy” (pdf). Retrieved 2008-01-07.[dead link]
- Schifitto G, Navia BA, Yiannoutsos CT, et al. (September 2007). “Memantine and HIV-associated cognitive impairment: a neuropsychological and proton magnetic resonance spectroscopy study”. AIDS 21 (14): 1877–86. doi:10.1097/QAD.0b013e32813384e8. PMID 17721095.
- Corbett J (September 2007). “Memantine/Gabapentin for the treatment of congenital nystagmus”. Curr Neurol Neurosci Rep 7 (5): 395–6. doi:10.1007/s11910-007-0061-z. PMID 17764629.
- Aman, Michael (Interviewee) (2010-07-29). Drug Used in Alzheimer’s Tested In Kids With Autism. Ohio: Ohio State University Medical Center.
- Borghol, Amne; Kirkwood A, Hawawini F (May 2010). “Memantine for the Treatment of Migraine”. US Pharm 35 (5): 28–35.
- Wang, R.; Zhang, D. (2005). “Memantine prolongs survival in an amyotrophic lateral sclerosis mouse model”. European Journal of Neuroscience 22 (9): 2376–2380. doi:10.1111/j.1460-9568.2005.04431.x. PMID 16262676.
- Costa, ACS; Boada R, Hutaff-Lee C, Schrader A, Weitzenkamp D, Benke TA, Goldson EJ (July 17, 2012). “Antagonism of NMDA receptors as a potential treatment for Down syndrome: a pilot randomized controlled trial”. Translational Psychiatry 2 (e141). doi:10.1038/tp.2012.66.

|
6-4-1981
|
Salts of dihalo-2-quinoxaline carboxylic acids, their preparation and pharmaceutical formulations containing them.
|
|
|
2-25-1981
|
Salts of dihalo-3,4-dihydro-3-oxo-2-quinoxaline carboxylic acids and hindered amines
|
|
|
10-25-1978
|
Drugs or medicines for influencing the central nervous system
|
|
12-16-2011
|
NOVEL THERAPEUTIC COMPOUNDS
|
|
|
12-16-2011
|
PROCESS FOR THE PREPARATION OF 1-BROMO-3,5-DIMETHYL ADAMANTANE
|
|
|
11-18-2011
|
PROCESS FOR PREPARING MEMANTINE
|
|
|
11-2-2011
|
CARNITINE CONJUGATES OF ADAMANTANAMINES DERIVATIVES AS DUAL PRODRUGS FOR VARIOUS USES
|
|
|
10-28-2011
|
PROCESS FOR MANUFACTURING ADAMANTANE DERVATIVES WITH HIGH YIELD
|
|
|
9-30-2011
|
Immediate release formulations of 1-aminocyclohexane compounds, memantine and neramexane
|
|
|
8-19-2011
|
Compositions, Methods, and Kits for Treating Influenza Viral Infections
|
|
|
7-13-2011
|
Carnitine Conjugates of Adamantanamines and Neramexane Derivatives as Dual Prodrugs for Various Uses
|
|
|
5-6-2011
|
SALTS OF MEMANTINE AND COX-INHIBITORS AND THEIR CRYSTAL FORM IN THE TREATMENT OF PAIN
|
|
|
5-4-2011
|
FLUORO-CONTAINING DERIVATIVES OF HYDROGENATED PYRIDO[4,3-B]INDOLES WITH NEUROPROTECTIVE AND COGNITION ENHANCING PROPERTIES, PROCESS FOR PREPARING, AND USE
|
Further reading
- Lipton SA (2005). “The molecular basis of memantine action in Alzheimer’s disease and other neurologic disorders: low-affinity, uncompetitive antagonism”. Current Alzheimer research 2 (2): 155–65. doi:10.2174/1567205053585846. PMID 15974913.
External links
Filed under: Uncategorized