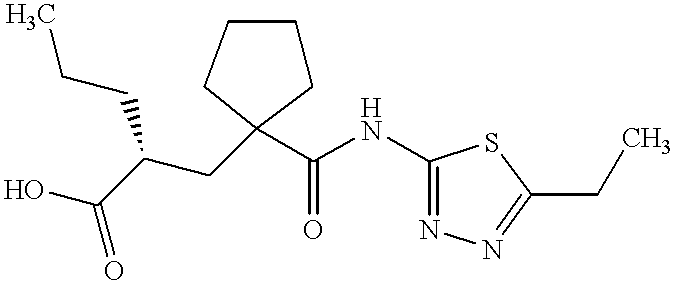
![UK-414,495 structure.png]()
UK-414,495
Molecular Formula: C16H25N3O3S
Molecular Weight: 339.453
UK 414495
CAS 388630-36-2
OF
(-)-(2R)-2-[[1-[[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl]cyclopentyl]methyl]pentanoic acid;
AND
Cyclopentanepropanoic acid, 1-[[(5-ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl]-α-propyl-, (αR)-
((R)-2-({1-[(5-ethyl-1,3,4-thiadiazol-2-yl) carbamoyl]cyclopentyl}methyl) valeric acid)
(2R)-2-[(1-{[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl}cyclopentyl) methyl]pentanoic acid
…………………………………………………
Cas 337962-93-3 RACEMIC…………2-[[1-[[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl]cyclopentyl]methyl]pentanoic acid
…………………………………………………………………..
ITS ENANTIOMER
(+)-(2S)-2-[[1-[[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl]cyclopentyl]methyl]pentanoic acid……………337962-74-0
Pfizer Inc.
CAS SUMMARY
| Cas number |
388630-36-2
337962-74-0 (enantiomer)
337962-93-3 (racemate)
388630-59-9 (sodium salt) |
![Figure US20020052370A1-20020502-C00058]() desired
desired
UK-414,495 is a drug developed by Pfizer for the treatment of female sexual arousal disorder.[1] UK-414,495 acts as a potent, selective inhibitor of the enzyme neutral endopeptidase, which normally serves to break down the neuropeptide VIP. The consequent increase in VIP activity alters blood flow to the genital region leading to increased lubrication and muscle relaxation.[2][3][4]
Female sexual arousal consists of a number of physiological responses resulting from increased genital blood. Vasoactive intestinal peptide (VIP), neuropeptide Y and to a lesser extent nitric oxide are neurotransmitters found in the vasculature of the genitalia. Neutral endopeptidase (NEP) modulates the activity of neuropeptides including VIP.
The aim of this study was to investigate the control of genital blood flow by VIP and endogenous neuropeptides using a selective NEP inhibitor [UK-414,495, ((R)-2-({1-[(5-ethyl-1,3,4-thiadiazol-2-yl) carbamoyl]cyclopentyl}methyl) valeric acid)].
![Chemical structure for SureCN5924982]()
A female equivalent of Viagra could soon be available to help women increase their sexual arousal, scientists claim.
For years they have endeavoured to create an alternative for women that mimics the effects of the male Viagra pill.
Now, the pharmaceutical company behind the original pill has created a prototype which increases blood flow to the genitalia in a similar way to Viagra.
Pfizer have come up with a prototype version of the female equivalent of Viagra
More than half of women experience sexual dysfunction at some point in their lives.
They may suffer a lack of desire, emotional or mental health problems and physical problems that mean they avoid having sex.
Pharmaceutical giant Pfizer has developed a drug, so far called only UK-414,495, which is supposed to increase sexual arousal, but will not affect desire, mood or emotional problems.
Some women take Viagra with mixed results and the drug has been used in fertility treatment to increase blood flow to the pelvis and encourage an embryo to implant in the womb.
But this is the first pill that claims to be an equivalent of the male Viagra.
The research, which involved animals, is published by the British Journal of Pharmacology, though Pfizer say they won’t develop the drug and warn that the chemical may not work the same way in humans, according to the Telegraph.
Chris Wayman, the lead researcher, said: ‘Before this work, we knew surprisingly little about the processes that control all of these changes.
Pfizer claim the tablets may help overcome female sexual arousal disorder
‘Now that we are beginning to establish the pathways involved in sexual arousal, scientists may be able to find ways of helping women who would like to overcome female sexual arousal disorder.
‘While the particular chemical compound in this research did not prove appropriate for further developments, the implications of the research could lead to the development of a product in the future.’
Viagra was originally developed as a treatment for high blood pressure and the heart condition angina, but men who took part in early trials realised the drug had an interesting side effect.
Clinical trials suggested the drug had little effect on angina and instead induced erections in men.
The drug first went on sale in 1998 and has since been prescribed to 25million men, creating a multi-billion pound global market.
The name Viagra has become so associated with men’s erectile problems that many cures are marketed as ‘herbal viagra’.
It is known by many nicknames, including Vitamin V and the Blue Pill.
Read more: http://www.dailymail.co.uk/health/article-1265842/Female-Viagra-help-women-increase-sexual-arousal.html#ixzz39lkmpSik
…………………………………
scheme
http://www.google.com/patents/US20020052370
![Figure US20020052370A1-20020502-C00014]()
![Figure US20020052370A1-20020502-C00015]()
![Figure US20020052370A1-20020502-C00017]()
![Figure US20020052370A1-20020502-C00018]()
Ex |
Prec |
n |
Y |
Data |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 43 |
Prep 37 |
0 |
|
1H NMR (CDCl3, 400 MHz) δ: 0.92 (t, 3H), 1.35 (t, 3H), 1.25-1.80 (m, 11H), 2.20-2.50 (m, 4H), 2.95 (q, 2H), 12.10 (bs, 1H); LRMS: m/z 339.8 (MH+) Anal. Found: C, 56.46; H, 7.46; N, 12.36. C16H25N3O3S requires C, 56.62; H, 7.44; N, 12.37%. |
Example 29 (2R)-2-[(1-{[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl}cyclopentyl) methyl]pentanoic acid
[0354]
![Figure US20020052370A1-20020502-C00058]() desired
desired[0355] and
Example 30 (2S)-2-[(1-{[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl}cyclopentyl) methyl]pentanoic acid
[0356]
![Figure US20020052370A1-20020502-C00059]() undesired
undesired[0357] The acid from Example 4 (824 mg) was further purified by HPLC using an AD column and using hexane:iso-propanol:trifluoroacetic acid (85:15:0.2) as eluant to give the title compound of example 29 as a white foam, 400 mg, 99.5% ee, 1H NMR (CDCl3, 400 MHz) δ: 0.90 (t, 3H), 1.36 (m, 6H), 1.50-1.80 (m, 9H), 2.19 (m, 1H), 2.30 (m, 1H), 2.44 (m, 1H), 2.60 (m, 1H), 2.98 (q, 2H), 12.10-12.30 (bs, 1H), LRMS: m/z 338 (MH−), [α]D=−9.0°(c=0.1, methanol),
and
the title compound of example 30 as a white foam, 386 mg, 99% ee, 1H NMR (CDCl3, 400 MHz) δ: 0.90 (t, 3H), 1.38 (m, 6H), 1.50-1.79 (m, 9H), 2.19 (m, 1H), 2.30 (m, 1H), 2.44 (m, 1H), 2.60 (m, 1H), 2.98 (q, 2H), 12.10-12.27 (bs, 1H);
[0358] LRMS: m/z 338 (MH−); and [α]D=+3.8°(c=0.1, methanol).
[0359] Alternatively, Example 29 may be prepared as follows:
[0360] To a solution of the product from Preparation 51a (574 g, 1.45 mol) in dichloromethane (2.87 L) was added trifluoroacetic acid (1.15 L) over a period of 50 minutes with cooling at 10° C. After addition was complete, the reaction was allowed to warm to ambient temperature with stirring under a nitrogen atmosphere for 24 hours. Deionised water (2.6 L) was then added. The reaction mixture was then washed with deionised water (3×2.6 L). The dichloromethane layer was concentrated to a volume of approximately 1 L to give the crude title compound (439 g, 1.29 mol, 96% yield) as a solution in dichloromethane. A purified sample of the title compound was obtained using the following procedure. To a dichloromethane solution (2.34 L) of the crude product, that had been filtered to remove any particulate contamination, was added isopropyl acetate (1.38 L). The resultant mixture was distilled at atmospheric pressure whilst being simultaneously replaced with isopropyl acetate until the solution temperature reached 87° C. The heating was stopped and the solution was allowed to cool to ambient temperature with stirring for 14 hours to give a cloudy brown solution. The agitation rate was then increased and crystallisation commenced. The suspension was then allowed to granulate for 12 hours at ambient temperature. The resultant suspension was then cooled to 0° C. for 3.5 hours and the solid was then collected by filtration. The filter cake was then washed with isopropyl acetate (2×185 ml, then 2×90 ml) and the solid was dried under vacuum at 40-45° C. for 18 hours to give the title compound (602 g, 0.18 mol, 70% yield) as a cream coloured, crystalline solid;
m.p.: 130-136° C.;
LRMS (negative APCI): m/z [M−H]− 338;
1H-NMR (CDCl3, 300 MHz) δ: 0.92 (t, 3H), 1.27-1.52 (m, 7H), 1.52-1.89 (m, 8H), 2.11-2.27 (m, 1H), 2.27-2.37 (m, 1H), 2.42-2.55 (m, 1H), 2.65 (dd, 2H), 3.00 (q, 2H), 12.25 (bs, 1H).
[0361] Example 29 may be purified as follows:
[0362] The title product from Example 29 was disolved in methanol. To this solution was added sodium methoxide (1 equivalent) in methanol (1 ml/g of Example 29) and the mixture was stirred at room temperature for 20 minutes. The solvent was removed in vacuo and the residue was azeotoped with ethyl acetate to give a brown residue. Ethyl acetate was added and the solution filtered to give a brown solid which was washed with tert-butylmethyl ether to give the crude sodium salt of Example 29. This crude product (35 g) was partitioned between water (200 ml) and ethyl acetate (350 ml). Concentrated hydrochloric acid (˜7 ml) was added until the pH of the aqueous layer was pH2. The aqueous phase was washed with ethyl acetate (2×100 ml). The combined layers were dried using magnesium sulphate. The solvent was removed in vacuo to give a light brown solid (31 g). Ethyl acetate (64 ml, 2 ml/g) and diisopropyl ether (155 ml, 5 ml/g) were added and the mixture heated to 68° C. until a clear solution was obtained (˜30 min). Upon cooling to room temperature, crystallisation of the free acid occurred. After 30 minutes stirring at room temperature the product was collected by filtration and washed with diisopropyl ether. The product was dried in a vacuum oven at 50° C. overnight. (20.2 g, 61% recovery from the sodium salt.); m.p. 135 degC (determined using a Perkin Elmer DSC7 at a heating rate of 20° C./minute).
[0372] The title compound of Example 29 metabolysed to form (2R)-1-(2-{[(5-ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl}pentyl)cyclopentanecarboxylic acid.
[0373] This compound was prepared as follows:
[0374] The product from Preparation 102 (430 mg, 1 mmol) was taken up in ethanol (5 mls) and methanol (1 ml) and hydrogenated at 30 psi hydrogen pressure at room temperature for 2 h. The mixture was then filtered through a plug of Arbocel®) and evaporated to a yellow oil. This oil was purified by column chromatography using firstly 19:1, then 9:1 DCM:MeOH as eluant to provide the product as a clear oil (120 mg, 35%); 1HNMR (400 MHz, CDCl3) 0.88 (t, 3H), 1.20-1.88 (m, 13H), 1.90-2.03 (m, 1H), 2.24-2.38 (m, 1H), 2.43-2.72 (m, 2H), 2.95 (q, 2H); LRMS m/z 340.2 (M+H).
Example 31 (R)-2-{[1-({[2-(Hydroxymethyl)-2,3-dihydro-1H-inden-2-yl]amino}carbonyl)-cyclopentyl]methyl}pentanoic acid
[0375] and
Example 32 (S)-2-{[1-({[2-(Hydroxymethyl)-2.3-dihydro-1H-inden-2-yl]amino}carbonyl)-cyclopentyl]methyl}pentanoic acid
[0376]
[0377] 2-{[1-({[2-(Hydroxymethyl)-2,3-dihydro-1H-inden-2-yl]amino}carbonyl)-cyclopentyl]methyl}pentanoic acid (WO 9110644, Example 8) was further purified by HPLC using an AD column and hexane:isopropanol:trifluoroacetic acid (90:10:0.1) as eluant, to give the title compound of Example 31, 99% ee, [α]D=+10.40 (c=0.067, ethanol) and the title compound of Example 32, 99% ee, [α]D=−10.9° (c=0.046, ethanol).
………………..
http://www.google.com/patents/US6734186
Example 7 (+)-2-[(1-{[(5-Ethyl-1,3,4-thiadiazol-2-yl)amino]carbonyl}cyclopentyl)methyl]pentanoic Acid (F63)
The acid from Preparation 18 (18/ex4) (824 mg) was further purified by HPLC using an AD column and using hexane:iso-propanol:trifluoroacetic acid (85:15:0.2) as eluant to give the title compound of example 7 as a white foam, 386 mg, 99% ee,1H NMR (CDCl3, 400 MHz) δ: 0.90 (t, 3H), 1.38 (m, 6H), 1.50-1.79 (m, 9H), 2.19 (m, 1H), 2.30 (m, 1H), 2.44 (m, 1H), 2.60 (m, 1H), 2.98 (q, 2H), 12.10-12.27 (bs, 1H); LRMS: m/z 338 (MH-); and [α]D=+3.80°(c=0.1, methanol)
……………………………………….
Novel selective inhibitors of neutral endopeptidase for the treatment of female sexual arousal disorder. Synthesis and activity of functionalized glutaramides
J Med Chem 2006, 49(14): 4409
David C. Pryde, Graham N. Maw, Simon Planken, Michelle Y. Platts, Vivienne Sanderson, Martin Corless, Alan Stobie, Christopher G. Barber, Rachel Russell, Laura Foster, Laura Barker, Christopher Wayman, Piet Van Der Graaf, Peter Stacey, Debbie Morren, Christopher Kohl, Kevin Beaumont, Sara Coggon, and Michael Tute
pp 4409–4424
Publication Date (Web): June 15, 2006 (Article)
DOI: 10.1021/jm060133g
Supporting Info
Female sexual arousal disorder (FSAD) is a highly prevalent sexual disorder affecting up to 40% of women. We describe herein our efforts to identify a selective neutral endopeptidase (NEP) inhibitor as a potential treatment for FSAD. The rationale for this approach, together with a description of the medicinal chemistry strategy, lead compounds, and SAR investigations are detailed. In particular, the strategy of starting with the clinically precedented selective NEP inhibitor, Candoxatrilat, and targeting low molecular weight and relatively polar mono-carboxylic acids is described. This led ultimately to the prototype development candidate R-13, for which detailed pharmacology and pharmacokinetic parameters are presented.
ACID ENTRY 13
δH(CDCl3, 400 MHz) 0.92 (3H, t), 1.35 (3H, t), 1.25-
1.80 (11H, m), 2.20-2.50 (4H, m), 2.95 (2H, q),
12.10 (1H, b s); MS
m/z
(TS+) 340 (M+H+
…………………………………………..
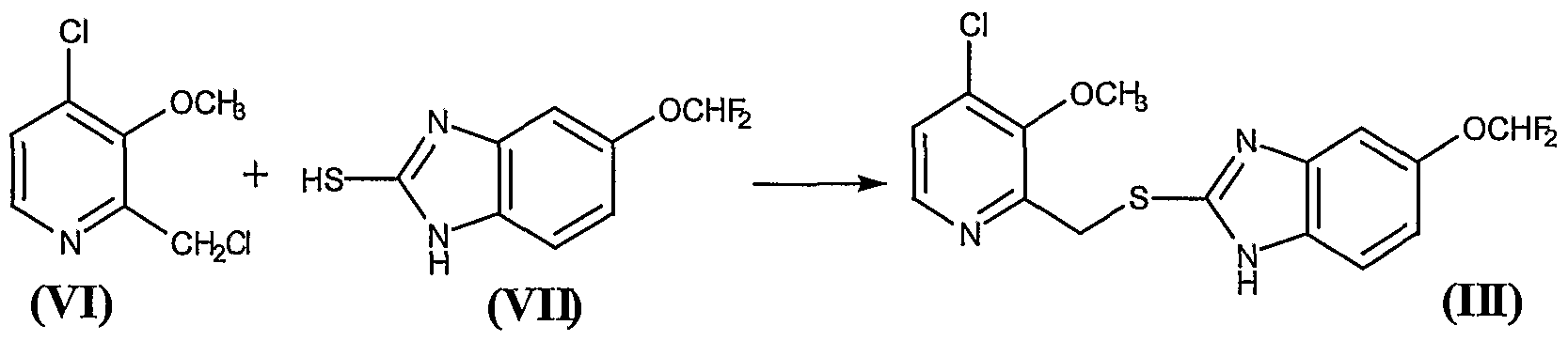
WO 2002002513
http://www.google.com/patents/WO2002002513A1?cl=en
…………………..
WO 2002003995
http://www.google.com/patents/WO2002003995A2?cl=en
Scheme 12
LiAIHψ THF, 6hr at reflux
![Figure imgf000030_0002]()
![Figure imgf000030_0003]()
![Figure imgf000030_0004]()
![Figure imgf000030_0005]()
Example 1
( f?)-2-r(1 r(5-ethyl-1.3.4-thiadiazol-2-yl)aminolcarbonyl)cvclopentyl) methyllpentanoic acid
and
Example 2
( S)-2-r(1-fr(5-Ethyl-1.3.4-thiadiazol-2-vnaminolcarbonyl)cvclopentyl)- methyllpentanoic acid
The title product from stage c) below (824mg) was further purified by HPLC using an AD column and using hexane:/sσ-propanol:trifluoroacetic acid (85:15:0.2) as elutant to give the title product from Example 1 , 400mg, 99.5% ee, 1H NMR (CDCI3, 400MHz) δ: 0.90 (t, 3H), 1.36 (m, 6H), 1.50-1.80 (m, 9H), 2.19 (m, 1 H), 2.30 (m, 1 H), 2.44 (m, 1 H), 2.60 (m, 1 H), 2.98 (q, 2H), 12.10-12.30 (bs, 1 H), LRMS : m/z 338 (MH“ ), [α]D = -9.0° (c = 0.1 , methanol), and the title product from Example 2, 386mg, 99% ee, 1H NMR (CDCl3, 400MHz) δ: 0.90 (t, 3H), 1.38 (m, 6H), 1.50-1.79 (m, 9H), 2.19 ( , 1 H), 2.30 ( , 1H), 2.44 (m, 1 H), 2.60 (m, 1 H), 2.98 (q, 2H), 12.10-12.27 (bs, 1H); LRMS: m/z 338 (MH“); and [α]D = +3.8° (c = 0.1 , methanol)
Preparation of Starting Materials a) 1 -r2-(tø/t-Butoxycarbonyl)-4-pentvπ-cvclopentane carboxylic acid
A mixture of 1 -[2-(tø t-butoxycarbonyl)-4-pentenyl]-cyclopentane carboxylic acid (EP 274234) (23g, 81.5mmol) and 10% palladium on charcoal (2g) in dry ethanol (200ml) was hydrogenated at 30psi and room temperature for 18 hours. The reaction mixture was filtered through Arbocel®, and the filtrate evaporated under reduced pressure to give a yellow oil. The crude product was purified by column chromatography on silica gel, using ethyl acetate:pentane (40:60) as the eluant, to provide the desired product as a clear oil, 21 g, 91%; 1H NMR (CDCI3, 0.86 (t, 3H), 1.22-1.58 (m, 15H), 1.64 (m, 4H), 1.78 (dd, 1H), 2.00-2.18 ( , 3H), 2.24 ( , 1H); LRMS : m/z 283 (M-HV b) tert-Butyl 2-1Ϊ1 -flT5-ethyl-1.3.4-thiadiazol-2-vnaminolcarbonyl)- cvclopentvDmethyllpentanoate.
1 -(3-Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (0.21 mmol), 1 – hydroxybenzotriazole hydrate (0.2mmol), N-methylmorpholine (0.31 mmol) and 2-amino-5-ethyl-1 ,3,4-thiadiazole (0.22mmol) were added to a solution of the product from stage a) above (150mg, 0.53mmol) in N,N- dimethylformamide (3ml), and the reaction stirred at 90°C for 18 hours. The cooled solution was diluted with ethyl acetate (90ml), washed with water
(3x25ml), and brine (25ml), then dried (MgSO ) and evaporated under reduced pressure. The crude product was purified by chromatography on silica gel, using ethyl acetate:pentane (30:70) as the eluant to afford the title compound, 92%; 1H NMR (CDCI3, 300MHz) δ: 0.82 (t, 3H), 1.20-1.80 (m, 22H), 1.84 (m, 1 H), 2.20 (m, 4H), 3.04 (q, 2H), 9.10 (bs, 1 H); LRMS : m/z
396.2 (MH+).
c) . 2-r(1-H,(5-ethyl-1.3.4-thiadiazol-2-yl)amino1carbonyl)cvclopentyl) methyllpentanoic acid.
Trifluoroacetic acid (5ml) was added to a solution of the title product from stage b) above (0.31 mmol) in dichloromethane (5ml), and the solution stirred at room temperature for 4 hours. The reaction mixture was concentrated under reduced pressure and the residue azeotroped with toluene and dichloromethane to afford the title compound as a clear oil, 81 %, 1H NMR
(CDCI3, 400MHz) δ: 0.92 (t, 3H), 1.35 (t, 3H), 1.25-1.80 (m, 11 H), 2.20-2.50 (m, 4H), 2.95 (q, 2H), 12.10 (bs, 1 H); LRMS : m/z 339.8 (MH+); Anal. Found: C, 56.46; H, 7.46; N, 12.36. Cι6H25N3O3S requires C, 56.62; H, 7.44; N, 12.37%.
………………………………………
Xiaoli Meng, James L. Maggs, David C. Pryde, Simon Planken, Rosalind E. Jenkins, Torren M. Peakman, Kevin Beaumont, Christopher Kohl, B. Kevin Park, and Andrew V. Stachulski
pp 6165–6176
Publication Date (Web): November 07, 2007 (Article)
DOI: 10.1021/jm0706766
Supporting Info
………………………………..
References
- ‘Female Viagra’ will help women increase their sexual arousal. Daily Mail Online 14th April 2010
- Armer R, Warne P, Witherington J (2006). “Recent disclosures of clinical drug candidates”. Drug News & Perspectives 19 (1): 65–72.PMID 16550257. ISSN 0214-0934
- Angulo, J. (2010). “Neutral endopeptidase inhibition: could it have a role in the treatment of female sexual arousal disorder?”. British Journal of Pharmacology 160: 48. doi:10.1111/j.1476-5381.2010.00693.x.
- Wayman, C.; Baxter, D.; Turner, L.; Van Der Graaf, P.; Naylor, A. (2010). “UK-414,495, a selective inhibitor of neutral endopeptidase, potentiates pelvic nerve-stimulated increases in female genital blood flow in the anaesthetized rabbit”. British Journal of Pharmacology160 (1): 51–59. doi:10.1111/j.1476-5381.2010.00691.x. PMC 2860206. PMID 20412068.
SEE
The discovery of small molecule inhibitors of neutral endopeptidase. Structure-activity studies on functionalized glutaramides
Chem Biol Drug Des 2006, 67(1): 74
Optimization of oral pharmacokinetics in the discovery of clinical candidates for the treatment of sexual dysfunction
237th ACS Natl Meet (March 22-26, Salt Lake City) 2009, Abst MEDI 173
Novel selective inhibitors of neutral endopeptidase for the treatment of female sexual arousal disorder. Synthesis and activity of functionalized glutaramides
J Med Chem 2006, 49(14): 4409
Bioorganic & Medicinal Chemistry (2007), 15(1), 142-159
Journal of Medicinal Chemistry (2007), 50(24), 6165-6176.
|
|
5-7-2004
|
Treatment of sexual dysfunction
|
|
|
11-15-2002
|
Treatment of sexual dysfunction
|
|
|
5-3-2002
|
Cyclopentyl-substituted glutaramide derivatives as inhibitors of neutral endopeptidase
|
| Citing Patent |
Filing date |
Publication date |
Applicant |
Title |
| US6734186 * |
Nov 8, 2000 |
May 11, 2004 |
Pfizer Inc. |
Phosphodiesterase 2 inhibitor |
| US7956195 * |
Dec 21, 2007 |
Jun 7, 2011 |
Abbott Laboratories |
reacting arylboronic acids with a cycloalkanone, in the presence of a rhodium catalyst or BINAP, to form a substituted arylcycloalkanone, then formin of a hydantoin, alkylation of the hydantoin, resolution, hydrolysis of the hydantoin to the amino acids and esterification of acids; chemical intermediates |
| WO2005007166A1 * |
Jul 12, 2004 |
Jan 27, 2005 |
Alasdair Mark Naylor |
Treatment of sexual dysfunction |
Female Sexual Response
The female sexual response phase of arousal is not easily distinguished from the phase of desire until physiological changes begin to take place in the vagina and clitoris as well as other sexual organs. Sexual excitement and pleasure are accompanied by a combination of vascular and neuromuscular events which lead to engorgement of the clitoris, labia and vaginal wall, increased vaginal lubrication and dilatation of the vaginal lumen (Levin, 1980; Ottesen, 1983; Levin, 1991; Levin, 1992; Sjoberg, 1992; Wagner, 1992; Schiavi et al., 1995; Masters et al., 1996; Berman et al., 1999).
Vaginal engorgement enables transudation to occur and this process is responsible for increased vaginal lubrication. Transudation allows a flow of plasma through the epithelium and onto the vaginal surface, the driving force for which is increased blood flow in the vaginal capillary bed during the aroused state. In addition engorgement leads to an increase in vaginal length and luminal diameter, especially in the distal ⅔ of the vaginal canal. The luminal dilatation of the vagina is due to a combination of smooth muscle relaxation of its wall and skeletal muscle relaxation of the pelvic floor muscles. Some sexual pain disorders such as vaginismus are thought to be due, at least in part, by inadequate relaxation preventing dilatation of the vagina; it has yet to be ascertained if this is primarily a smooth or skeletal muscle problem. (Levin, 1980; Oltesen, 1983; Levin, 1991; Levin, 1992; Sjoberg, 1992; Wagner, 1992; Schiavi et al., 1995; Master et al., 1996; Berman et al., 1999).
The vasculature and micro vasculature of the vagina are innervated by nerves containing neuropeptides and other neurotransmitter candidates. These include calcitonin gene-related peptide (CGRP), neuropeptide Y (NPY; Sequence No. 4), nitric oxide synthase (NOS), substance P and vasoactive intestinal peptide (VIP; Sequence No. 8) (Hoyle et al., 1996). Peptides that are present in the clitoris are discussed infra. Nitric oxide synthase, which is often colocalised with VIP (Sequence No. 8), displays a greater expression, immunologically, in the deep arteries and veins rather than in the blood vessels of the propria (Hoyle et al., 1996).
Female Sexual Dysfunction
It is known that some individuals can suffer from female sexual dysfunction (FSD). FSD is best defined as the difficulty or inability of a woman to find satisfaction in sexual expression. FSD is a collective term for several diverse female sexual disorders (Leiblum, 1998, Berman et al., 1999). The woman may have lack of desire, difficulty with arousal or orgasm, pain with intercourse or a combination of these problems. Several types of disease, medications, injuries or psychological problems can cause FSD.
Studies investigating sexual dysfunction in couples reveals that up to 76% of women have complaints of sexual dysfunction and that 30-50% of women in the USA experience FSD.
Sub-types of FSD include hypoactive sexual desire disorder, female sexual arousal disorder, orgasmic disorder and sexual desire disorder.
Treatments in development are targeted to treat specific subtypes of FSD, predominantly desire and arousal disorders.
The categories of FSD are best defined by contrasting them to the phases of normal female sexual response: desire, arousal and orgasm (Leiblum 1998). Desire or libido is the drive for sexual expression—and manifestations often include sexual thoughts either when in the company of an interested partner or when exposed to other erotic stimuli. In contrast, sexual arousal is the vascular response to sexual stimulation, an important component of which is vaginal lubrication and elongation of the vagina. Thus, sexual arousal, in contrast to sexual desire, is a response relating to genital (e.g. vaginal and clitoral) blood flow and not necessarily sensitivity. Orgasm is the release of sexual tension that has culminated during arousal. Hence, FSD typically occurs when a woman has an inadequate or unsatisfactory response in any of these phases, usually desire, arousal or orgasm. FSD categories include hypoactive sexual desire disorder, sexual arousal disorder, orgasmic disorders and sexual pain disorders.
Hypoactive sexual desire disorder is present if a woman has no or little desire to be sexual, and has no or few sexual thoughts or fantasies. This type of FSD can be caused by low testosterone levels, due either to natural menopause or to surgical menopause. Other causes include illness, medications, fatigue, depression and anxiety.
Female sexual arousal disorder (FSAD) is characterised by inadequate genital response to sexual stimulation. The genitalia (e.g. the vagina and/or the clitoris) do not undergo the engorgement that characterises normal sexual arousal. The vaginal walls are poorly lubricated, so that intercourse is painful. Orgasms may be impeded. Arousal disorder can be caused by reduced oestrogen at menopause or after childbirth and during lactation, as well as by illnesses, with vascular components such as diabetes and atherosclerosis. Other causes result from treatment with diuretics, antihistamines, antidepressants eg SSRIs or antihypertensive agents. FSAD is discussed in more detail infra.
Sexual pain disorders (which include dyspareunia and vaginismus) are characterised by pain resulting from penetration and may be caused by medications which reduce lubrication, endometriosis, pelvic inflammatory disease, inflammatory bowel disease or urinary tract problems.
The prevalence of FSD is difficult to gauge because the term covers several types of problem, some of which are difficult to measure, and because the interest in treating FSD is relatively recent. Many women’s sexual problems are associated either directly with the female ageing process or with chronic illnesses such as diabetes and hypertension.
There are wide variations in the reported incidence and prevalence of FSD, in part explained by the use of differing evaluation criteria, but most investigators report that a significant proportion of otherwise healthy women have symptoms of one or more of the FSD subgroups. By way of example, studies comparing sexual dysfunction in couples reveal that 63% of women had arousal or orgasmic dysfunction compared with 40% of men have erectile or ejaculatory dysfunction (Frank et al., 1978).
However, the prevalence of female sexual arousal disorder varies considerably from survey to survey. In a recent National Health and Social Life Survey 19% of women reported lubrication difficulties whereas 14% of women in an outpatient gynaecological clinic reported similar difficulties with lubrication (Rosen et al., 1993).
Several studies have also reported dysfunction with sexual arousal in diabetic women (up to 47%), this included reduced vaginal lubrication (Wincze et al., 1993). There was no association between neuropathy and sexual dysfunction.
Numerous studies have also shown that between 11-48% of women overall may have reduced sexual desire with age. Similarly, between 11-50% of women report problems with arousal and lubrication, and therefore experience pain with intercourse. Vaginismus is far less common, affecting approximately 1% of women.
Studies of sexually experienced women have detailed that 5-10% have primary anorgasmia. Another 10% have infrequent orgasms and a further 10% experience them inconsistently (Spector et al., 1990).
Because FSD consists of several subtypes that express symptoms in separate phases of the sexual response cycle, there is not a single therapy. Current treatment of FSD focuses principally on psychological or relationship issues. Treatment of FSD is gradually evolving as more clinical and basic science studies are dedicated to the investigation of this medical problem. Female sexual complaints are not all psychological in pathophysiology, especially for those individuals who may have a component of vasculogenic dysfunction (eg FSAD) contributing to the overall female sexual complaint. There are at present no drugs licensed for the treatment of FSD. Empirical drug therapy includes oestrogen administration (topically or as hormone replacement therapy), androgens or mood-altering drugs such as buspirone or trazodone. These treatment options are often unsatisfactory due to low efficacy or unacceptable side effects.
Since interest is relatively recent in treating FSD pharmacologically, therapy consists of the following:- psychological counselling, over-the-counter sexual lubricants, and investigational candidates, including drugs approved for other conditions. These medications consist of hormonal agents, either testosterone or combinations of oestrogen and testosterone and more recently vascular drugs, that have proved effective in male erectile dysfunction. None of these agents has been demonstrated to be very effective in treating FSD.
Female Sexual Arousal Disorder (FSAD)
The sexual arousal response consists of vasocongestion in the pelvis, vaginal lubrication and expansion and swelling of the external genitalia. The disturbance causes marked distress and/or interpersonal difficulty. Studies investigating sexual dysfunction in couples reveals that there is a large number of females who suffer from sexual arousal dysfunction; otherwise known as female sexual arousal disorder (FSAD).
The Diagnostic and Statistical Manual (DSM) IV of the American Psychiatric Association defines Female Sexual Arousal Disorder (FSAD) as being:
“a persistent or recurrent inability to attain or to maintain until completion of the sexual activity adequate lubrication-swelling response of sexual excitement. The disturbance must cause marked distress or interpersonal difficulty.”
FSAD is a highly prevalent sexual disorder affecting pre-, peri- and post menopausal (±HRT) women. It is associated with concomitant disorders such as depression, cardiovascular diseases, diabetes and UG disorders.
The primary consequences of FSAD are lack of engorgement/swelling, lack of lubrication and lack of pleasurable genital sensation. The secondary consequences of FSAD are reduced sexual desire, pain during intercourse and difficulty in achieving an orgasm.
It has recently been hypothesised that there is a vascular basis for at least a proportion of patients with symptoms of FSAD (Goldstein et al., 1998) with animal data supporting this view (Park et al., 1997).
Drug candidates for treating FSAD, which are under investigation for efficacy, are primarily erectile dysfunction therapies that promote circulation to the male genitalia. They consist of two types of formulation, oral or sublingual medications (Apomorphine, Phentolamine, Sildenafil), and prostaglandin (PGE1-Alprostadil) that are injected or administered transurethrally in men, and topically to the genitalia in women.
The present invention seeks to provide an effective means of treating FSD, and in particular FSAD.
SUMMARY
The present invention is based on findings that FSAD is associated with reduced genital blood flow—in particular reduced blood flow in the vagina and/or the clitoris. Hence, treatment of women with FSAD can be achieved by enhancement of genital blood flow with vasoactive agents. In our studies, we have shown that cAMP mediates vaginal and clitoral vasorelaxation and that genital (e.g. vaginal and clitoral) blood flow can be enhanced/potentiated by elevation of cAMP levels. This is a seminal finding.
In this respect, no one has previously proposed that FSAD can be treated in such a way—i.e. by direct or indirect elevation of cAMP levels. Moreover, there are no teachings in the art to suggest that FSAD was associated with a detrimental modulation of cAMP activity and/or levels or that cAMP is responsible for mediating vaginal and clitoral vasorelaxation. Hence, the present invention is even further surprising.
In addition, we have found that by using agents of the present invention it is possible to increase genital engorgement and treat FSAD—e.g. increased lubrication in the vagina and increased sensitivity in the vagina and clitoris. Thus, in a broad aspect, the present invention relates to the use of a cAMP potentiator to treat FSD, in particular FSAD.
The present invention is advantageous as it provides a means for restoring a normal sexual arousal response—namely increased genital blood flow leading to vaginal, clitoral and labial engorgement. This will result in increased vaginal lubrication via plasma transudation, increased vaginal compliance and increased genital (e.g. vaginal and clitoral) sensitivity. Hence, the present invention provides a means to restore, or potentiate, the normal sexual arousal response.
More particularly, the present invention relates to:
A pharmaceutical composition for use (or when in use) in the treatment of FSD, in particular FSAD; the pharmaceutical composition comprising an agent capable of potentiating cAMP in the sexual genitalia of a female suffering from FSD, in particular FSAD; wherein the agent is optionally admixed with a pharmaceutically acceptable carrier, diluent or excipient.
The use of an agent in the manufacture of a medicament (such as a pharmaceutical composition) for the treatment of FSD, in particular FSAD; wherein the agent is capable of potentiating cAMP in the sexual genitalia of a female suffering from FSD, in particular FSAD.
A method of treating a female suffering from FSD, in particular FSAD; the method comprising delivering to the female an agent that is capable of potentiating cAMP in the sexual genitalia; wherein the agent is in an amount to cause potentiation of cAMP in the sexual genitalia of the female; wherein the agent is optionally admixed with a pharmaceutically acceptable carrier, diluent or excipient.
An assay method for identifying an agent that can be used to treat FSD, in particular FSAD, the assay method comprising: determining whether an agent can directly or indirectly potentiate cAMP; wherein a potentiation of cAMP in the presence of the agent is indicative that the agent may be useful in the treatment of FSD, in particular FSAD.
Filed under:
sex arousal Tagged:
495,
FEMALE VIAGRA,
PFIZER,
sex arousal,
sexual arousal,
sexual dysfunction,
UK 414,
uk 414.495,
uk 495 ![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()







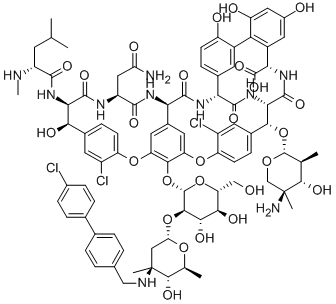 oritavancin
oritavancin oritavancin
oritavancin
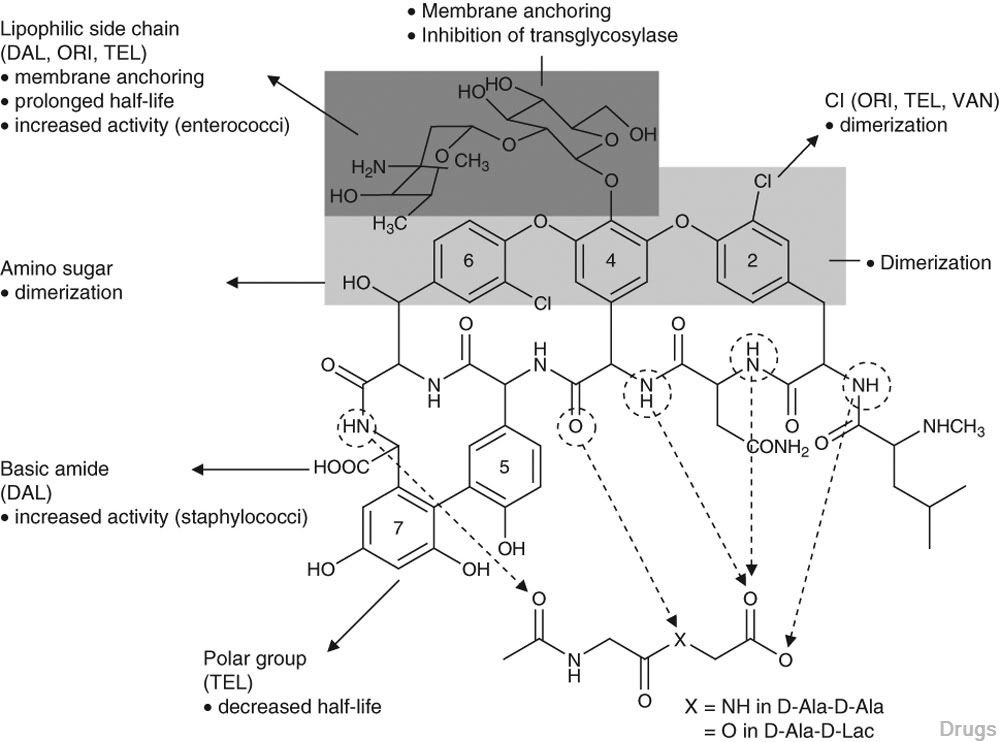
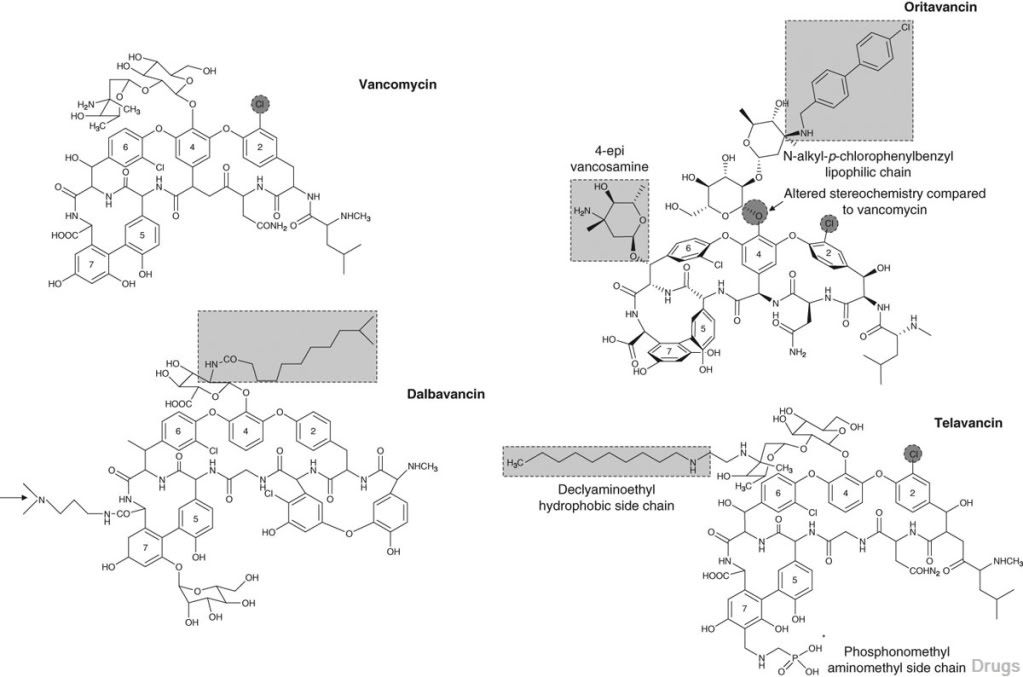



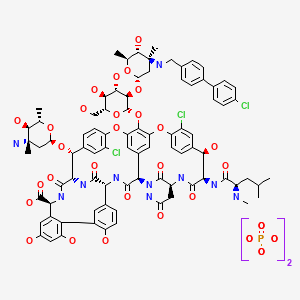 ORITAVANCIN DIPHOSPHATE
ORITAVANCIN DIPHOSPHATE

















































 and
and  or of their enantiomers, followed by crystallization of the corresponding cis-benzoates, (2S,4R)-18 or(2S,4S)-18, from which (+)- or (−)-1 were obtained as described for (±)-1. The ee’s of (+)- and (−)-ketoconazole were determined by HPLC on the CSP Chiralcel OD-H.
or of their enantiomers, followed by crystallization of the corresponding cis-benzoates, (2S,4R)-18 or(2S,4S)-18, from which (+)- or (−)-1 were obtained as described for (±)-1. The ee’s of (+)- and (−)-ketoconazole were determined by HPLC on the CSP Chiralcel OD-H.