CL-315555/CL-315585
CL-318952
CV-001
Verteporfin (trade name Visudyne), a benzoporphyrin derivative, is a medication used as a photosensitizer for photodynamic therapy to eliminate the abnormal blood vessels in the eye associated with conditions such as the wet form of macular degeneration. Verteporfin accumulates in these abnormal blood vessels and, when stimulated by nonthermal red light with awavelength of 693 nm in the presence of oxygen, produces highly reactive short-lived singlet oxygen and other reactive oxygen radicals, resulting in local damage to the endothelium and blockage of the vessels.[1][2]
Verteporfin is also used off-label for the treatment of central serous retinopathy.[3]

Verteporfin, otherwise known as benzoporphyrin derivative (trade name Visudyne®), is a medication used as a photosensitizer for photodynamic therapy to eliminate the abnormal blood vessels in the eye associated with conditions such as the wet form of macular degeneration. Verteporfin accumulates in these abnormal blood vessels and, when stimulated by nonthermal red light with a wavelength of 693 nm in the presence of oxygen, produces highly reactive short-lived singlet oxygen and other reactive oxygen radicals, resulting in local damage to the endothelium and blockage of the vessels.
Administration
Verteporfin is given intravenously, 15 minutes before laser treatment.[1]

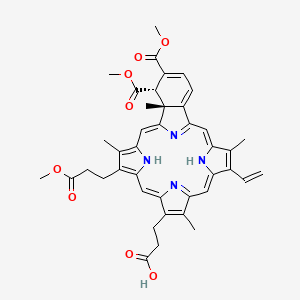
VISUDYNE® (verteporfin) for Injection is a light activated drug used inphotodynamic therapy. The finished drug product is a lyophilized dark green cake. Verteporfin is a 1:1 mixture of two regioisomers (I and II), represented by the following structures:
![VISUDYNE®<br />
(verteporfin) Structural Formula Illustration” /></p>
<p>The chemical names for the verteporfin regioisomers are:</p>
<p>9-methyl (I) and 13-methyl (II) trans-(±)-18-ethenyl-4,4a-dihydro-3,4-bis(methoxycarbonyl)-4a,8,14,19-tetramethyl-23H, 25H-benzo[<i>b</i>]porphine-9,13-dipropanoate</p>
<p>The molecular formula is C<sub>41</sub>H<sub>42</sub>N<sub>4</sub>O<sub>8</sub> with a molecular weight of approximately 718.8. Each mL of reconstituted VISUDYNE contains:</p>
<table class=](http://images.rxlist.com/images/rxlist/verteporfin1.gif)
Contraindications
Side effects
Most common side effects are blurred vision, headache, and local effects at the injection site. Also, photosensitivity; it is advised to avoid exposure to sunlight and unscreened lighting until 48 hours after the injection of verteporfin.[1]
Interactions
None known. Verteporfin has no influence on the liver enzyme CYP3A4, which metabolises many pharmaceutical drugs.[1]
…………………..
http://www.google.com/patents/WO2011017809A1?cl=en
Verteporfin (CAS # 129497-78-5) is a benzoporphyrin derivative which has been used clinically for photodynamic therapy of age related macular degeneration (23).
Verteporfin is photoactivated for photodynamic therapy to eliminate the abnormal blood vessels in the eye associated with conditions such as the wet form of macular
degeneration. Verteporfin accumulates in these abnormal blood vessels and, when stimulated by nonthermal red light with a wavelength of 693 ran in the presence of oxygen, the photoactivated verteporfin produces highly reactive short-lived singlet oxygen and other reactive oxygen radicals, resulting in local damage to the endothelium and blockage of the vessels. Benzoporphoryrins, are described for example, in US patents 5,095,030, 5,214,036, and 6,008,241.
Verteporfin (CAS # 129497-78-5) as used herein may include the two regioisomers as shown below:
and the 2 entantiomers of each of the two regioisomers as shown below:
The verteporfin as disclosed herein contains at least one chiral center and thus may exist in various stereoisomeric forms. If desired, such stereoisomers, including enantiomers, may be separated using techniques standard in the art (for example, chiral columns). However, racemic mixtures or mixtures containing more than one diastereomer may also be used and are contenplated herein. However, the compounds tested herein were in either of the trans entantiomers shown above. The compounds shown in Formulas IA, IB, Tables 1, 2 and Figure 10, are representative of the individual optical isomers, enantiomers or diastereomers as the case may be, as well as mixtures of these individual chiral isomers.
Visudyne™, as used herein, is the liposomal formulation of verteporfin used in humans for photodynamic therapy. Visudyne™ is given intravenously, usually within 15 minutes prior to laser treatment to eliminate the abnormal blood vessels in the eye in the treatment of wet macular degeneration. The verteporfin compound accumulates in these abnormal blood vessels and, when stimulated by a nonthermal red light laser with a wavelength of 693 nm in the presence of oxygen, produces highly reactive short-lived singlet oxygen and other reactive oxygen radicals, resulting in local damage to the endothelium and blockage of the vessels. Patients given Visudyne™ experience photosensitivity and are advised to avoid exposure to sunlight and unscreened lighting for at least 48 hours after the injection of verteporfin.
In contrast to the current use of verteporfin in photodynamic therapy, subjects administered the BPDs described herein, in accordance with the methods and uses described herein, do not require photoactivation of the BPD via nonthermal red light laser with a wavelength of 693 nm or otherwise. The activity of the BPDs to inhibit early stage autophagy is independent of the activity associated with photoactivation and would likely be hindered by photoactivation. Accordingly, a person of skill in the art would appreciate that the precautions associated with photosensitivity should also apply to the present methods and uses (i.e. avoid exposure to sunlight and unscreened lighting for at least 48 hours after the injection of of the BPD).
References
- Verteporfin monograph
- Scott, L. J.; Goa, K. L. (2000). “Verteporfin”. Drugs & aging 16 (2): 139–146; discussion 146–8. doi:10.2165/00002512-200016020-00005. PMID 10755329.
- Adelman, R.; Adelman, R. A. (2013). “Profile of verteporfin and its potential for the treatment of central serous chorioretinopathy”.Clinical Ophthalmology 7: 1867–1875. doi:10.2147/OPTH.S32177. PMC 3788817. PMID 24092965.
External links
| Patent | Submitted | Granted |
|---|---|---|
| Photodynamic therapy for the treatment of hair loss [US7090691] | 2004-01-22 | 2006-08-15 |
| PHOTOTHERAPY METHODS AND DEVICES COMPRISING EMISSIVE ARYL-HETEROARYL COMPOUNDS [US2012015998] | 2012-01-19 | |
| PHOTOTHERAPY DEVICES AND METHODS COMPRISING SUBSTITUTED CARBAZOLE COMPOUNDS [US2012016449] | 2012-01-19 | |
| PHOTODYNAMIC THERAPY FOR THE TREATMENT OF HAIR LOSS [US2008056996] | 2008-03-06 | |
| FACTOR VII CONJUGATES FOR SELECTIVELY TREATING NEOVASCULARIZATION DISORDERS [US2008206227] | 2008-08-28 | |
| Factor VII conjugates for selectively treating neovascularization disorders [US2006052286] | 2006-03-09 | |
| Methods of treating neuralgic pain [US2004220167] | 2004-11-04 |
| Systematic (IUPAC) name | |
|---|---|
| 3-[(23S,24R)-14-ethenyl-5-(3-methoxy-3-oxopropyl)-22,23-bis(methoxycarbonyl)-4,10,15,24-tetramethyl-25,26,27,28-tetraazahexacyclo[16.6.1.13,6.18,11.113,16.019,24]octacosa-1,3,5,7,9,11(27),12,14,16,18(25),19,21-dodecaen-9-yl]propanoic acid | |
| Clinical data | |
| Trade names | Visudyne |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a607060 |
| Pregnancy cat. |
|
| Legal status | |
| Routes | Intravenous |
| Identifiers | |
| CAS number | 129497-78-5  |
| ATC code | S01LA01 |
| PubChem | CID 5362420 |
| DrugBank | DB00460 |
| ChemSpider | 21106402  |
| UNII | 0X9PA28K43  |
| KEGG | D01162  |
| ChEBI | CHEBI:60775  |
| ChEMBL | CHEMBL2218885  |
| Chemical data | |
| Formula | C41H42N4O8 |
| Molecular mass | 718.794 g/mol |
Filed under: Uncategorized Tagged: photodynamic therapy, Verteporfin